Use the standard reduction potentials (Appendix M) for the half-reactions [AuCl 4 ] (aq) + 3
Question:
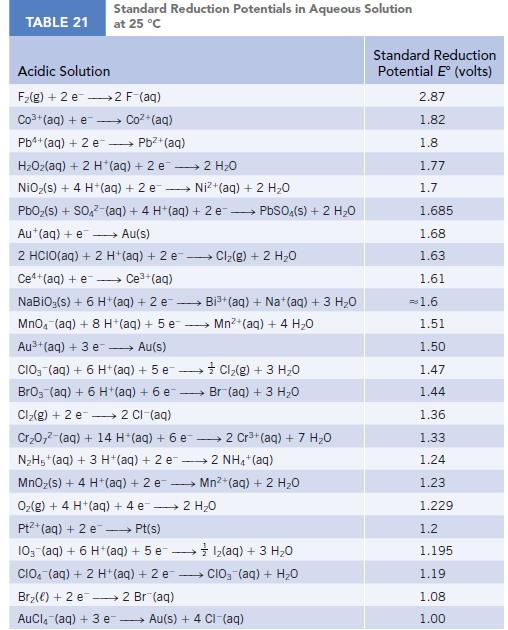
Use the standard reduction potentials (Appendix M) for the half-reactions [AuCl4]−(aq) + 3 e− → Au(s) + 4 Cl−(aq) and Au3+(aq) + 3 e− → Au(s) to calculate the value of Kformation for the complex ion [AuCl4]−(aq).
Data given in Appendix M
Transcribed Image Text:
TABLE 21 Standard Reduction Potentials in Aqueous Solution at 25 °C Acidic Solution F₂(g) + 2 e 2 F-(aq) Co3+ (aq) + e Coz+(aq) Pb4+ (aq) + 2 e - Pb²+(aq) HzOz(aq) + 2 H*(aq) +2e → 2H2O NiO₂(s) + 4 H+ (aq) + 2 e→→→→→→ Ni+(aq) + 2 HO PbO₂ (s) + SO4² (aq) + 4 H+ (aq) + 2e → PbSO4(s) + 2 H₂O Au+ (aq) + e→→→→→ Au(s) 2 HCIO(aq) + 2 H+ (aq) + 2 e-- - Ce+(aq) + e→→→→ Ce³+ (aq) NaBiO;(s) + 6 H+ (aq) + 2 e- → MnO4 (aq) + 8 H+ (aq) + 5 e Au³+ (aq) + 3 e→→→→→ Au(s) CIO (aq) + 6 H+ (aq) +5 e→→→→→→ BrO3 (aq) + 6 H+ (aq) + 6 e- Cl₂(g) + 2 e 2 Cl-(aq) Cr₂0,² (aq) + 14 H*(aq) + 6 e 2 Cr³+ (aq) + 7 H₂O N₂H5+ (aq) + 3 H+ (aq) + 2 e2 NH4+ (aq) MnO₂ (s) + 4 H+ (aq) + 2 e O₂(g) + 4 H+(aq) + 4 e 2 H₂O Pt+ (aq) + 2 e →→→→→ Pt(s) 10- (aq) + 6 H+ (aq) + 5 e1₂(aq) + 3 H₂O CIO (aq) + 2 H+ (aq) + 2 e CIO₂ (aq) + H₂O Br₂() +2 e 2 Br(aq) AuCl(aq) + 3 e → Cl₂(g) + 2 H₂O → Bi³+ (aq) + Na+ (aq) + 3 H₂O Mn²+ (aq) + 4H₂O Cl₂(g) + 3 H₂O Br (aq) + 3 H₂O Mn²+ (aq) + 2 H₂O Au(s) + 4 CI-(aq) Standard Reduction Potential E (volts) 2.87 1.82 1.8 1.77 1.7 1.685 1.68 1.63 1.61 = 1.6 1.51 1.50 1.47 1.44 1.36 1.33 1.24 1.23 1.229 1.2 1.195 1.19 1.08 1.00
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To calculate the formation constant Kf for the complex ion AuCl4aq we need to use the following equa...View the full answer

Answered By

Vineet Kumar Yadav
I am a biotech engineer and cleared jee exam 2 times and also i am a math tutor. topper comunity , chegg India, vedantu doubt expert( solving doubt for iit jee student on the online doubt solving app in live chat with student)
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Use the standard reduction potentials (Appendix M) for the half-reactions [Zn(OH) 4 ] 2 (aq) + 2 e Zn (s) + 4 OH (aq) and Zn 2+ (aq) + 2e Zn(s) to calculate the value of K formation for the...
-
Use the standard reduction potentials to find the equilibrium constant for each of the following reactions at 25°C: (a) (b) (c) Br2(1) + 21-(aq )- 2Br_(aq) + 12(s) 5Fe2 + (aq) +MnO4 (aq ) + 8H +...
-
Use the standard reduction potentials (Appendix M) for the half-reactions Hg 2 Cl 2 (s) + 2e 2 Hg() + 2 Cl (aq) and Hg 2 2+ (aq) + 2 e 2 Hg() to calculate the value of K sp for Hg 2 Cl 2 . Data...
-
Raheem & Co. purchased a fixed asset on 1.4.2018 for Rs.2,50,000. Depreciation is to be provided @10% annually according to the Straight-line method. The books are closed on 31st March every year....
-
A project has estimated annual net cash flows of $150,000. It is estimated to cost $885,000. Determine the cash payback period.
-
Marx Industries had the following transactions. 1. Borrowed $5,000 from the bank by signing a note. 2. Paid $3,100 cash for a computer. 3. Purchased $850 of supplies on account. Instructions (a)...
-
During 2016, The Alberta Oil & Gas Company began an exploration project in Montana. The company had paid \($500,000\) for the drilling rights on a tract of 500 acres of land. The company then spent...
-
Pellegrino Watch Company reported the following income statement data for a 2-year period. Pellegrino uses a periodic inventory system. The inventories at January 1, 2011, and December 31, 2012, are...
-
If $15,500 is deposited in an account earning interest compounded continuously at 4.00% for 12 years, what is the effective rate of interest? Format answer rounded to 3 decimal places AS A PERCENT....
-
In 1937, R. Schwartz and M. Schmiesser prepared a yellow-orange bromine oxide (BrO 2 ) by treating Br 2 with ozone in a fluorocarbon solvent. Many years later, J. Pascal found that, on heating, this...
-
The reaction occurring in the cell in which Al 2 O 3 and aluminum salts are electrolyzed is Al 3+ (aq) +3 e Al(s). If the electrolysis cell operates at 5.0 V and 1.0 10 5 A, what mass of aluminum...
-
A 1.35-kg object is attached to a horizontal spring of force constant 2.5 N/cm. The object is started oscillating by pulling it 6.0 cm from its equilibrium position and releasing it so that it is...
-
Weighted average and FIFO in process costing. Which method is more used in practice and why?
-
What important role Pareto Charts play in operations quality management both in domestic and global markets
-
David is the wealthiest resident in his neighborhood. Some of his poorer neighbors have hacked his wireless internet so they can save money for more important things like food and health insurance....
-
what are the benefits and challenges of creating and using budgets.
-
Webstercompany produces 25,000 units of product A, 20,000 units of product B and 10,000 units of product C from same manufacturing process at cost of $340,000. Other Details: Product A & B = Joint...
-
An electric utility company recently issued $25 million of mandatory redeemable preferred stock that is redeemable in 10 years. In its audit, the IRS wishes to classify the preferred stock as debt....
-
r = 0.18 Find the coefficients of determination and non-determination and explain the meaning of each.
-
This problem concerns the design of a divider for unsigned binary numbers that will divide a 16-bit dividend by an 8-bit divisor to give an 8-bit quotient. Assume that the start signal (ST = 1) is 1...
-
Design a 4 Ã 4 keypad scanner for the following keypad layout. (a) Assuming only one key can be pressed at a time, find the equations for a number decoder given R 3-0 and C 3- 0 , whose output...
-
Four pushbuttons (B 0 , B 1 , B 2 , and B 3 ) are used as inputs to a logic circuit. Whenever a button is pushed, it is debounced, after which the circuit loads the button number in binary into a...
-
Good day tutor, could you kindly assist in answering the questions (1-3 only) b elow. Please take note of the instruction and data set showns below. The first data set has salaries of recent...
-
If the future value of an ordinary, 6-year annuity is $5,600 and interest rates are 7.5 percent, what is the future value of the same annuity due
-
To operate practically as a substitute for cash or a credit device, a negotiable instrument must be Question 21 options: a) conditional without the risk of being collectable. b) qualified with a...

Study smarter with the SolutionInn App


