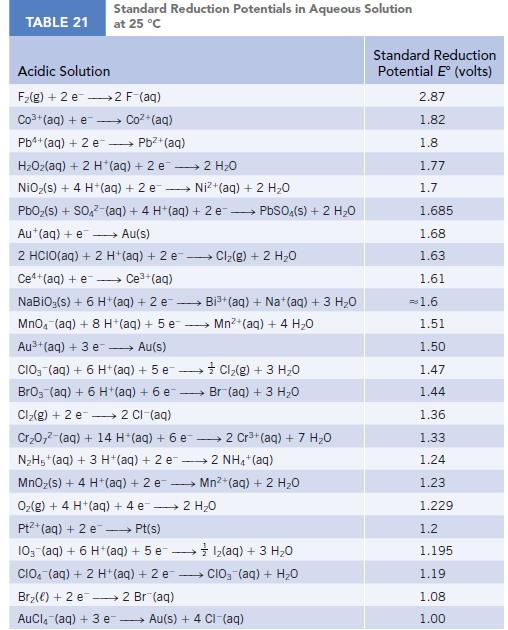
Use the table of standard reduction potentials (Appendix M) to calculate r G for the following
Question:
Use the table of standard reduction potentials (Appendix M) to calculate ΔrG° for the following reactions at 298 K.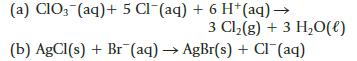
Data given in Appendix M
Transcribed Image Text:
(a) CIO3(aq) + 5 Cl(aq) + 6 H+ (aq) → 3 Cl₂(g) + 3 H₂O(l) (b) AgCl(s) + Br (aq) → AgBr(s) + Cl(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a The relation between Gibbs free energy and ...View the full answer

Answered By

Benish Ahmad
I'm a professional software engineer. I'm lectutrer at GCUF and I have 3 years of teaching experience. I'm looking forward to getting mostly computer science work including:
Programming fundamentals
Object oriented programming
Data structures
object oriented design and analysis
Database system
Computer networks
Discrete mathematics
Web application
I am expert in different computer languages such as C++, java, JavaScript, Sql, CSS, Python and C#. I'm also have excellent knowledge of essay writing and research. I have worked in other Freelancing website such as Fiverr and Upwork. Now I have finally decided to join the SolutionInn platform to continue with my explicit work of helping dear clients and students to achieve their academic dreams. I deliver plagiarism free work and exceptional projects on time. I am capable of working under high pressure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Use the table of standard reduction potentials (Appendix M) to calculate r G for the following reactions at 298 K. Data given in Appendix M (a) 3 Cu(s) + 2NO3(aq) + 8 H+ (aq) 3 Cu+ (aq) + 2 NO(g) +...
-
Using the standard reduction potentials listed in Appendix E, calculate the equilibrium constant for each of the following reactions at 298 K: (a) Fe(s) + Ni2+(aq) Fe2+ (aq) + Ni(s) (b) Co(s) + 2 H+...
-
Using the standard reduction potentials listed in Appendix E, calculate the equilibrium constant for each of the following reactions at 298 K: (a) Cu(s) + 2 Ag+ (aq) Cu2+ (aq) + 2 Ag(s) (b) 3 Ce4+...
-
Built-Tite uses job order costing. The T-account below summarizes Factory overhead activity for the current year. Factory Overhead Debit Credit 16,200 106,600 25,200 60,200 1. Compute total applied...
-
Southern Rail Inc. is considering acquiring equipment at a cost of $442,500. The equipment has an estimated life of 10 years and no residual value. It is expected to provide yearly net cash flows of...
-
Match each ratio or percentage with its formula by entering the appropriate letter for each numbereditem. Ratios or Percentages 1. Current ratio 2. Net profit margin 3. Inventory turnover ratio 4....
-
The basic concept behind a learning organization is: A. The popularity to change. B. The people change. C. The adaptation to change. D. The resources of change.
-
Megerle Custom Cabinet Co. uses the job order cost system. In recording payroll transactions, the following accounts are used: Cash ................ Administrative Salaries Wages Payable ...............
-
This document is beneficial because it facilitates a bidding process that may drive down prices or increase value as vendors compete to win the bid. Question 7 options: Service Level Agreement (SLA)...
-
In 2005, global SO 2 emission was estimated to be 12.83 Gg (gigagrams). According to the EPA, 71% of SO 2 emissions into the atmosphere is from coalfired power plants. How much coal (in metric tons)...
-
Calculate equilibrium constants for the following reactions at 298 K. Indicate whether the equilibrium as written is reactant- or product-favored at equilibrium. (a) 2 Cl(aq) + Br(e) (b) Fe+ (aq) +...
-
Bendt Displays designs and manufactures displays used in mobile devices. Serious flooding throughout North Carolina affected Bendt Displays facilities. Inventory was completely ruined, and the...
-
The following are several figures reported for Allister and Barone as of December 31, 2021: Inventory Sales Investment income Cost of goods sold Operating expenses Allister Barone $ 490,000 $290,000...
-
In which situations, if any, would it be appropriate to use GPS when employees are 'off the clock"? The case discusses various GPS devices, but some companies in the U.S. and Europe have started...
-
Donald Transport assembles prestige manufactured homes. Its job-costing system has two direct-cost categories (direct materials and direct manufacturing labor) and one indirect-cost pool...
-
Will AI threaten jobs in Accounting or Finance? Which functions will be the safest from being replaced by AI? Which functions are the most threatened? As an employee, how do you stay safe in the fast...
-
b. The interest payment on June 30, Year 2, and the amortization of the bond premium, using the straight-line method. Round to the nearest dollar. 3. Determine the total interest expense for Year 1....
-
Assume the same facts as in Exercise 5. But now you expect your tax rate on ordinary income to decline to 20% in year 3. How do your answers in Exercise 5 change? Would you make the Section 83(b)...
-
1. Which of the four major types of information systems do you think is the most valuable to an organization? 2. How do you critically associate the ideas of business agility and business efficiency...
-
Write a Verilog description of the following combinational circuit using concurrent statements. Each gate has a 5-ns delay, excluding the inverter, which has a 2-ns delay. C - ABCDAR
-
(a) Write Verilog code for a full subtracter using logic equations. (b) Write Verilog code for a 4-bit subtracter using the module defined in (a) as a component.
-
Write Verilog code for the following circuit. Assume that the gate delays arenegligible. (a) Using concurrent statements. (b) Using an always block with sequential statements. No latches should be...
-
ACCOUNTING HELP College Coasters is a San Diego-based merchandiser specializing in logo-adorned drink coasters. The company reported the following balances in its unadjusted trial balance at December...
-
Joliet Plumbing collected the following information on its business segments: Residential Division Commercial Division Operating Income $93,000 $260,000 Invested Assets $72,000 $190,000 Sales...
-
For the California Earned Income Tax Credit a taxpayer may be eligible to receive this credit at what age if all other qualifications are met

Study smarter with the SolutionInn App


