A 25.0-mL sample of 1.44 M NH 3 is titrated with 1.50 M HCl. Calculate the pH
Question:
A 25.0-mL sample of 1.44 M NH3 is titrated with 1.50 M HCl. Calculate the pH at the equivalence point.
Choose an indicator from Table 16.4, and justify your choice.
Table 16.4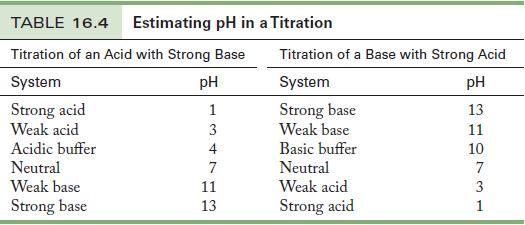
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
pH at the e...View the full answer

Answered By

Arun kumar
made more than four thousand assignments
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
A 10.0-mL solution of 0.300 M NH3 is titrated with a 0.100 M HCl solution. Calculate the pH after the following additions of the HCl solution: (a) 0.0 mL, (b) 10.0 mL, (c) 20.0 mL, (d) 30.0 mL, (e)...
-
Calculate the pH at the halfway point and at the equiva-lence point for each of the following titrations. a. 100.0 mL of 0.10 M HC7H5O2 (Ka = 6.4 105) titrated with 0.10 M NaOH b. 100.0 mL of 0.10 M...
-
Please write detailed roadmap/solution for all questions below. 1) An output of nmap search is shown below, a) Type the required terminal command and required parameters to obtain the shown output....
-
How do recruitment and selection practices contribute to high performance in an organization?
-
Predict the products of the following nucleophilic acyl substitutionreactions: (b) (a) NaOH NH3 H20 H (d) (c) Na* "OCH3 C CH3NH2 SCH3 "CH
-
We return once more to the native wild man to point out the influence which the reestablishment of the time dimension in the mythology of the Golden Age had upon opinions about him. It was in the...
-
Change \(46.113 \times 10^{8}\) by moving the decimal four places to the left.
-
Cretin Enterprises uses a predetermined overhead rate of $21.40 per direct labor-hour. This predetermined rate was based on 8,000 estimated direct labor-hours and $171,200 of estimated total...
-
A stock is expected to return 9% in a normal economy, 13% if theeconomy booms, and lose 5% if the economy moves into a recessionaryperiod. Economists predict a 60% chance of a normal economy, a 11% 2...
-
Exactly 50 mL of a 0.0500 M solution of ethylamine, a base with K b = 1.1 10 -6 , is titrated with 0.100 M HNO 3 . What is the pH at the equivalence point? Suggest a good indicator from Table 16.4...
-
Chloropropionic acid, ClCH 2 CH 2 COOH, is a weak monoprotic acid with Ka = 7.94 10 -5 . Calculate the pH at the equivalence point in a titration of 10.00 mL of 0.100 M chloropropionic acid with...
-
Bettys Grocery Store has three departmentsmeat, canned food, and produceeach of which has its own manager. All departments are housed in a single store. Recently, the produce department has been...
-
Given an array of bird sightings where every element represents a bird type id, determine the id of the most frequently sighted type. If more than 1 type has been spotted that maximum amount, return...
-
Consider the following. f(x) = 2x4 - - 8x +4 Find all relative extrema. (If an answer does not exist, enter DNE.) relative maximum (x, y) = relative minimum (x, y, = Find the points of inflection....
-
You are given a linked list consisting of just 0s, 1s and 2s. You need to return the sorted linked list. Code structure is already given, you need to add the remaining code to finish it. Input...
-
As you've learned, it is important to be able to determine the elements of a crime and there are several places to turn for assistance in doing so. First Look at the statute for the crime. For...
-
In the following exercises, express the limits as integrals. 60. 00 lim. av Ax over 11,3 61. 19 m5-7-1/JA over 10. 21 62. 69 lim 63. lim "M= "M= sin (2x; )Ax over [0: 1] cos(27x7 Ax over 10.11
-
What is e-commerce? What activities does it include? How do online transaction sites use databases and shopping cart software? Why is security critical for e-commerce success? Which protocol supports...
-
A copper rod of length L =18.0 in is to be twisted by torques T (see figure) until the angle of rotation between the ends of the rod is 3.08. (a) If the allowable shear strain in the copper is 0.0006...
-
What size of standard hydraulic copper tube from Appendix G.2 is required to transfer 0.06 m 3 /s of water at 80C from a heater where the pressure is 150 kPa to an open tank? The water flows from the...
-
Determine the required size of new Schedule 80 steel pipe to carry water at 160F with a maximum pressure drop of 10 psi per 1000 ft when the flow rate is 0.5 ft 3 /s.
-
A device designed to allow cleaning of walls and windows on the second floor of homes is similar to the system shown in Fig. 11.20. Determine the velocity of flow from the nozzle if the pressure at...
-
What happens when a signal whose wavelength is large relative to a barrier, encounters that barrier? Group of answer choices significant loss of energy on the far side of the barrier no diffraction...
-
The magnetic field around a bar magnet can be modeled by drawing __________. Group of answer choices curved lines from the north pole of the magnet to the south pole circular lines that form closed...
-
A(n) ______________________ is a scale in which the reference level changes with frequency. Group of answer choices intensity level sound pressure level reference weighted scale

Study smarter with the SolutionInn App


