Calculate H , S , and G for each of the following reactions at 298 K.
Question:
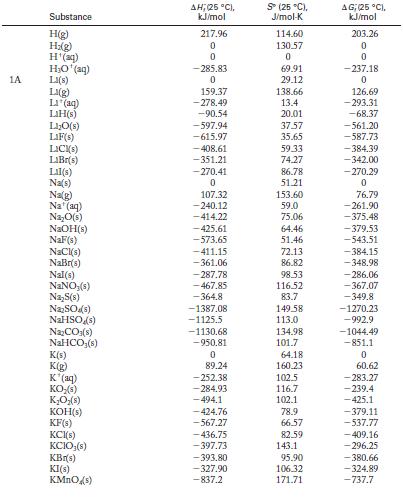
Calculate ΔH °, ΔS °, and ΔG ° for each of the following reactions at 298 K. State whether the direction of spontaneous reaction is consistent with the sign of the enthalpy change, the entropy change, or both. Use Appendix G for data.![]()
Appendix G
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To calculate the standard Gibbs free energy change AG standard enthalpy change AH and standard entro...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Calculate H , S , and G for each of the following reactions at 298 K. State whether the direction of spontaneous reaction is consistent with the sign of the enthalpy change, the entropy change, or...
-
Use data in Table 4H.1 or Appendix 2A to calculate the standard reaction entropy for each of the following reactions at 25C. For each reaction, interpret the sign and magnitude of the reaction...
-
Calculate the standard enthalpy, entropy, and Gibbs free energy for each of the following reactions at 298 K by using data in Appendix 2A. For each case, confirm that the value obtained from the...
-
Instructions: Read the footnotes included in the financial statements for H & B Bakery, then answer the following questions. *The exact requirement of this question, is to read the statements below...
-
What is the irradiation at surfaces A 2 , A 3 , and A 4 of Example 12.1 due to emission from A 1 ?
-
What does it mean to "fix" nitrogen, and what natural processes accomplish nitrogen fixation?
-
8. YOU BE THE JUDGE WRITING PROBLEM Cellwave was a limited partnership that applied to the Federal Communications Commission (FCC) for a license to operate cellular telephone systems. After the FCC...
-
The Diamond Glitter Company is in the process of preparing its financial statements for 2012. Assume that no entries for depreciation have been recorded in 2012. The following information related to...
-
Beginning inventory, purchases, and sales data for tennis rackets are as follows: 20 April 3 Inventory @ $13 units 17 11 Purchase @ $15 units 24 14 Sale units 9 21 Purchase @ $20 units 25 Sale 11...
-
Calculate H , S , and G for each of the following reactions. State whether the direction of spontaneous reaction is consistent with the sign of the enthalpy change, the entropy change, or both. Use...
-
Calculate G for the following reactions two diff erent ways: (1) Use Hesss law and the standard Gibbs free energies of formation, (2) Use G = H - TS. Compare the two values and judge whether you get...
-
Our sun rotates in a circular orbit about the center of the Milky Way galaxy. The radius of the orbit is 2.2 1020 m, and the angular speed of the sun is 1.1 10-15 rad/s. How long (in years) does it...
-
Describe the role that marketing research plays at Dunkin' Donuts. What sort of information does Dunkin' Donuts consider when it's determining where to locate a new store? Discuss the importance of...
-
(a) First perform a multiple regression with all the variables, what can you say about the significance of the variables based on only the p-values. Next use the "step" function to perform backward...
-
After reading Hamblin's article, you'll notice he uses all three rhetorical appeals to change his reader's approach to sleep. write brief paragraph (5 sentences) identifying the ONE rhetorical appeal...
-
Delta-Gamma-Vega neutral. Q5. On MARCH 1, a portfolio manager expects a market down turn in the 5.1 5.2 next 4 months and decides to use the JUN puts on the S&P500 index in order to protect the...
-
Discuss how ICT can create value in your business. Consider the various matters that might have impacted the existing ICT system during Covid-19 in the light of the case study. KPMG. (2022). KPMG...
-
Create a clustered bar chart using Severe, Minor, and Total Defects columns.
-
A survey of 70 college freshmen asked whether students planned to take biology, chemistry, or physics during their first year. Use the diagram to answer each question. How many of the surveyed...
-
A cell phone battery is rated at 3.85 V and can store 10.78 watt-hours of energy. (a) How much average current can it deliver over a period of 3 hours if it is fully discharged at the end of that...
-
A 1.2-kW toaster takes roughly 4 minutes to heat four slices of bread. Find the cost of operating the toaster twice per day for 2 weeks (14 days). Assume energy costs 9 cents/kWh.
-
A utility company charges 8.2 cents/kWh. If a consumer operates a 60-W light bulb continuously for one day, how much is the consumer charged?
-
When direct materials are requisitioned, they flow directly into (D) A. cost of goods sold account. B. finished goods inventory account. C. manufacturing overhead account. D. work in process...
-
Question 7 4 pts Find the slope of the line tangent to the graph of the function at the given value of x. y= (7/x) square root of (x) x = 4 (11/16) (11/16) (3/16) (3/16) Question 8 Find an equation...
-
1.Using the HCPCS code book, assign code(s) for the following scenario: Newborn was sent home with a Pediatric crib, hospital grade, fully enclosed. List the HCPCS code verified in the Tabular List...

Study smarter with the SolutionInn App


