State which compound in each of the following pairs you expect to have the higher boiling point.
Question:
State which compound in each of the following pairs you expect to have the higher boiling point. Explain your answer.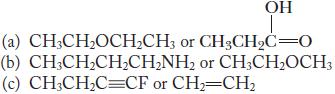
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a CH 3 CH 2 COOH hydrogen b...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
State which compound in each of the following pairs you expect to have the higher boiling point. Explain your answer.
-
Which compound in each of the following pairs would have the higher boiling point? Explain your answers. (a) (b) (c) (d) (e) (f) (g) (h) Hexane, CH3(CH2)4CH3, or nonane, CH3(CH2)7CH3 (i) OH or HO OH...
-
Did Hopkins materially misrepresent his health when applying for insurance? Does Golden Rule have the right to rescind his insurance policies?
-
Let the sequence [x (n T)) denote die input applied to a tapped-delay-line equalizer. Show char inter-symbol interference is eliminated completely by the equalizer provided that its frequency...
-
I suspect that most of us can view and appreciate most sports such as baseball, football, and basketball. What if you were to view a Cricket World Cup game? If you didn't know the rules you probably...
-
Consider the following \(3 \times 3\) game. (a) Identify all pairs of strategies where one strategy weakly dominates the other. (b) Assume you are allowed to remove a weakly dominated strategy of...
-
Paula Judge owns Judge Creative Designs. The trial balance of the firm for January 31, 2016, the first month of operations, is shown below. INSTRUCTIONS 1. Complete the worksheet for the month. 2....
-
. ident cation: 1. Under what circumstances or conditions may a partnership be dissolved? 2. Distinguish buying an interest in a partnership from investing in a partnership. 3. When a new partner...
-
Write the structural formula and name the organic product expected from the acid-catalyzed condensation reaction of CH 3 OH.
-
Draw all the isomers of trichlorobenzene.
-
Discuss what corporate governance is and why it is important to shareholders.
-
Why is cultural awareness important? List five different types of diversity. Which types of diversity will you most often have to consider in the workplace? Describe the role of leaders and managers...
-
Does forensic science have sufficient safeguards in place to avoid misuse of evidence in court situations?Why or why not?
-
Below is the taxpayer information for Sharon and Robert Dolan. They have two children who lived with them, David ( SS# 1 5 4 - 1 3 - 5 8 2 6 ) , age 1 3 ( date of birth: October 1 1 , 2 0 1 0 ) ,...
-
Claire Stevens is 29 years old and single, lives in an apartment, and has one child dependent that lives with her full time from a prior relationship. Last year she earned $118,000 as a market...
-
Consider the situation shown in Figure 3. A 1000V rms source delivers power to a load. The load consumes 100 kW with a power factor of 25 percent lagging. a) Find the phasor I, assuming that the...
-
Compare the source of capital processes to sales processes in terms of: a. The frequency of transactions b. The volume of transactions c. The magnitude in dollars of a single transaction d. The...
-
For each of the following transactions, indicate whether it increases, decreases, or has no effect on the following financial ratios: current ratio, debt-to-equity ratio, profit margin ratio, and...
-
An optical filter can be described by a Jones matrix Obtain the form of the emerging light for each of the following incident beams: (a) A plane polarized beam polarized at angle θ to...
-
Keeping Eq. (8.33) in mind, write an expression for the randomly polarized flux density component (I n ) of a partially polarized beam in terms of the Stokes parameters. To check your result, add a...
-
Construct a Mueller matrix for an isotropic plate of absorbing material having an amplitude transmission coefficient of t. What Mueller matrix will completely depolarize any wave without affecting...
-
2. If a company's assets are $100,000, liabilities are $50,000, and equity is $50,000, what is the company's debt-to- equity ratio? a) 0.5 b) 1 c) 2 d) 1.5 3. Which accounting principle requires that...
-
A river habitat rehabilitation is being considered. If a BCA is to be undertaken which or the following should be part of/included in the accounting stance? Group of answer choices a discount rate...
-
about the burger king one risk in each of the 4 ERM risk categories. Out of those risks you will choose one to do a risk assessment on as explained in Chapter 3. And finally, you will describe how...

Study smarter with the SolutionInn App


