What are the products of each of the following acidbase reactions? Indicate the acid and its conjugate
Question:
What are the products of each of the following acidbase reactions? Indicate the acid and its conjugate base, and the base and its conjugate acid.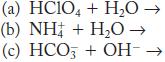
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a Solution HClO4 H2O C1 H3O Acid Bas...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
What are the products of each of the following acidbase reactions? Indicate the acid and its conjugate base and the base and its conjugate acid. (a) HNO 3 + H 2 O (b) HSO 4 + H 2 O (c) H 3 O + + F
-
What are the products of each of the following acidbase reactions? Indicate the acid and its conjugate base and the base and its conjugate acid. (a) HClO 4 + H 2 O (b) NH 4 + + H 2 O (c) HCO 3 + OH
-
You continue to use the buffer that you prepared in Example 6G.1 but are worried about how adding sodium hydroxide to the buffer solution will affect the pH, which could upset your experiment....
-
A) Suppose you wanted to make a photoconductor that interacts in the visible, green light range (500-570 nm). Which of the following semiconductors would be your best choice, and why? Si, AIP, InAs,...
-
For a senior project, a student was given the assignment to design a heat exchanger that meets the following specifications: Like many real-world situations, the customer hasn't revealed, or doesn't...
-
What is the relationship between (the probability of committing a Type II error) and the power of the test?
-
Question: Edo applied for insurance with Geico. In calculating his premium, Geico looked at his credit history and his financial circumstances. It did not offer him the best possible premium but this...
-
Alpha Airlines has ordered a new fleet of DC- 717s. At this stage of the contract, Alpha's operations manager must specify the seating configuration on the aircraft that will be used on the...
-
pleasw answer correctlu The Rosa model of Mohave Corporation is currently manufactured as a very plain umbrella with no decoration. The company is considering changing this product to a much more...
-
Write an equation to describe the proton transfer that occurs when each of these acids is added to water.
-
The following species react in aqueous solution. Predict the products, identify the acids and bases (and their conjugate species), and show the proton transfer in the acidbase reactions. (a) Ammonia...
-
Gorimapa Manufacturing Company produces desktop calendars. The following are the budgeted data for 3,000 units of Joystick produced in September 2020: Required During the period 2,800 were produced...
-
Explain what conflicts can arise within the firm between stockholders and managers, and between stockholders and bondholders, and discuss the techniques that firms can use to mitigate these potential...
-
WeGrow Inc. stock beta is 2.3. The one-year Treasury bill ($100 face value, risk-free) currently trades for $97.0874. The expected market return is 7%. a. What is the expected return of WeGrow's...
-
AEE Company manufactures a product that passes in four departments in a continuous process. Department C had no beginning work in process and transferred in 36,000 units from Department B, each with...
-
JoeCo, a coffee company, has generated $100mm in last twelve months ("LTM") Revenue and this figure is expected to grow $10mm annually. JoeCo's LTM EBITDA was $20mm and its EBITDA margin should...
-
How Would You Judge If A Remote Server Is Running Iis Or Apache? What Is Data Protection In Transit Vs Data Protection At Rest? You See A User Logging In As Root To Perform Basic Functions. Is This A...
-
What is the lex talionis view of punishment? How does it differ from the proportional view?
-
A bubble-point liquid feed is to be distilled as shown in Figure. Use the Edmister group method to estimate the mole-fraction compositions of the distillate and bottoms. Assume initial overhead and...
-
Why was electromagnetic induction discovered much earlier than its converse, the production of a magnetic field by a changing electric field?
-
Why are light waves able to travel through a vacuum whereas sound waves cannot?
-
A double star consists of two nearby stars that revolve around their center of mass. How can an astronomer recognize a double star from the characteristic frequencies of the light that reaches him...
-
1. Provide an explanation on Audit of Performance Information in the Public Sector.(Advanced Audit and Assurance) 2. What areSocial and Environmental Issues.(Advanced Audit and Assurance)
-
9 On January 1, 2024, Riverbed Company acquired all the assets and assumed all the liabilities of Oriole Company and merged Oriole into Riverbed. In exchange for the net assets of Oriole, Riverbed...
-
Part I. Recording the transactions. Exercise 1. Blue Co engaged in the following transactions during July 2022: Required: Based on the information below, journalize the transactions. (36 Points) July...

Study smarter with the SolutionInn App


