What is H at 25 C and 1.00 atm for the combustion of 1 mol ethane if
Question:
What is ΔH at 25 °C and 1.00 atm for the combustion of 1 mol ethane if ΔE = -1553 kJ?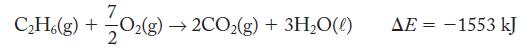
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
AH...View the full answer

Answered By

AJIN KURIAKOSE
I HAVE ELECTRONICS ENGINEERING DEGREE..AND MY AREA OF INTEREST IS MATHEMATICS,CONTROL SYSTEM,NETWORK,DIGITAL
4.70+
21+ Reviews
32+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
How much heat, in kilojoules, is evolved in the complete combustion of (a) 1.325 g C 4 H 10 (g) at 25 C and 1 atm; (b) 28.4 L C 4 H 10 (g) at STP; (c) 12.6 L C 4 H 10 (g) at 23.6 C and 738 mmHg?...
-
The difference in the standard free energies of formation for 1-butene and 2-methylpropene is 13.4 kJ mol 1 (3.2 kcal mol 1 ). (See the previous problem for a definition of G f .) (a) Which compound...
-
One way to evaluate fuels with respect to global warming is to determine how much heat they release during combustion relative to how much CO 2 they produce. The greater the heat relative to the...
-
The Sellinger Business School's Information Technology Service (ITS) is considering a new process to refurbish older computers in order to save on costs of buying new computers. The five steps to the...
-
One step in the gluconeogenesis pathway for the biosynthesis of glucose is the partial reduction of 3-phosphoglycerate to give glyceraldehydes 3-phosphate. The process occurs by phosphorylation with...
-
Geoworld Enterprises plc has the following information extracted from its statement of income and payroll systems:...
-
On January 1, 2016, Safetyway Group,a LO1 U.S. company, formed a Swiss subsidiary, TEurope AG. The subsidiary issued all of its currently outstanding common stock on that date. Selected accounts from...
-
Below are selected T-accounts for the RunnerTech Company. Below are selected T-accounts for the RunnerTech Company. Required: Complete the following journal entries and answer the following...
-
2. (16 points) A portfolio manager has a mandate to maintain the portfolio duration at 6 - 7 years. Currently, in his portfolio, half of them is investing in Bond A and half in Bond B. Bond A ...
-
Determine the entropy change when 1 mol H 2 O freezes at its normal freezing point of 0.0 C. Th e heat of fusion of water is 6.01 kJ/mol.
-
A 7.56-g sample of gas is in a balloon that has a volume of 10.5 L. Under an external pressure of 1.05 atm, the balloon expands to a volume of 15.00 L. Then the gas is heated from 0.0 C to 25.0 C. If...
-
A sample of radioactive material is initially found to have an activity of 115.0 decays/min. After 4 d 5 h, its activity is measured to be 73.5 decays/min. (a) Calculate the half-life of the...
-
A drive sprocket has 24 teeth and rotates at 20 pm. The driven sprocket has only 12 teeth. How many revolutions will the driven sprocket make per minute
-
Laila made the following property transfers. Payment to MSU for 15,000 daughter's tuition Payment to MSU for 12,000 daughter's room & board Donation to local Fire 1,000 Dept Payment to the doctor for...
-
An initial principal is to be quadrupled in eighteen years. Under the simple interest method and the compound interest method, what should be the annual rates that allow the capital to be quadrupled?...
-
Find the differentials for g(x)= (t^2 + 3t + 2)(t+4)
-
(a) Determine the torque that can be applied to a solid shaft of 50-mm diameter without exceeding an allowable shearing stress of 45 MPa. (b) Solve Part a, assuming that the solid shaft has been...
-
It is possible the robots will be so useful that the company needs to assign one for every employee, instead of sharing them. Change the number of robots required so that all 100 employees get their...
-
A Bloomberg Businessweek subscriber study asked, In the past 12 months, when traveling for business, what type of airline ticket did you purchase most often? A second question asked if the type of...
-
A horse galloped a mile in 2 min 35 s. What was its average speed in km/h?
-
Figure P.4.95 is a plot of n I and n R versus λ for a common metal. Identify the metal by comparing its characteristics with those considered in the chapter and discuss its optical...
-
Convert 0.296 cm 3 /s to m 3 /s.
-
_________________ establishes "the measurement of economic activity, time when such measurements are to be made and recorded, disclosures surrounding this activity, and preparation and presentation...
-
All of the following are sources of information that need to be specified when documenting client information, EXCEPT: Group of answer choices Client's report Personal observation Personal assumption...
-
Defined benefit plans are regarded by employers as volatile because when interest rates are low, the plan is more likely to be underfunded and employer contributions are more likely to be increased;...

Study smarter with the SolutionInn App


