What is the sign of w for the following processes if they occur at constant pressure? Consider
Question:
What is the sign of w for the following processes if they occur at constant pressure? Consider only PV work from gases and assume that all gases behave ideally.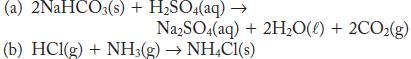
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a w is ...View the full answer

Answered By

Muhammad Salman Alvi
Well, I am a student of Electrical Engineeing from Information Technology University of Punjab. Just getting into my final year. I have always been good at doing Mathematics, Physics, hardware and technical subjects. Teaching profession requires a alot of responsibilities and challenges.
My teaching experience started as an home tutor a year ago. When I started teaching mathematics and physic subjects to an O Level student. He was about 14 years old. His name was Ibrahim and I used to teach him for about 2 hours daily. Teaching him required a lot of patience but I had to be polite with him. I used to give him a 5 min break after 1 hour session. He was quite weak in basic maths and calculation. He used to do quite a lot of mistakes in his homework which I gave him weekly. So I decided to teach him basics from scratch. He used to say that he got the concept even if he didn't. So I had to ask him again and again. I worked on his basics for a month and after that I started taking a weekly test sesions. After few months he started to improve gradually. Now after teaching him for about a year I can proudly say that he has improved alot. The most important thing was he managed to communicate all the difficullties he was facing. He was quite capable and patient. I had a sincere desire to help him reach to its full potential. So I managed to do that. We had a very good honest relationship of a student and a teacher. I loved teaching him as a tutor. Now having an experience of one year teaching I can read students quite well. I look forward to work as an online tutor who could help students in solving their all sort of difficulties, problems and queries.
4.90+
29+ Reviews
43+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
What is the sign of w for the following processes if they occur at constant pressure? Consider only PV work from gases, and assume that all gases behave ideally.
-
Calculate w for the following reactions that occur at 298 K and 1 atm pressure. Consider only PV work from the change in volume of gas, and assume that the gases are ideal and the chemical equation...
-
Calculate w for the following reactions that occur at 298 K and 1 atm pressure. Consider only PV work from the change in volume of gas, and assume that the gases are ideal and the chemical equation...
-
Recession is defined as... a) increase in unemployment b) decrease in consumer spending c) two consecutive quarters of negative economic growth d) both A & B
-
For a given a hydrogen atom to be acidic, the C?H bond must be parallel to the p orbital?s of the C=O bond (that is, perpendicular to the plane of the adjacent carbonyl group). Identify the most...
-
Use the Internet or Strayer Library to explore the relationship between information management and data storage techniques. Write a paper in which you: Ascertain the importance of information...
-
Question: When Theodore Staats went to his company's "Council of Honor Con- vention," he was accompanied by a woman who was not his wife although he told everyone she was. The company fired him....
-
Palisade Creek Co. is a merchandising business that uses the perpetual inventory system. The account balances for Palisade Creek Co. as of May 1, 2019 (unless otherwise indicated), are as follows:...
-
Time: 1/1/2020-12/31/2021 Frequency: Monthly Portfolio: 20% CRO-USD, 35% AAPL, 30% TSLA, 15% CMG Question: Beta and Cost of Equity for Each Company? RM=^GSPC Rf= ^IRX Please complete the Following...
-
Calculate w for the following reactions that occur at 298 K and 1 atm pressure. Consider only PV work from the change in volume of gas, and assume that the gases are ideal and the chemical equation...
-
When NaCl dissolves in water, can you accurately predict the sign of H for the dissolution of the soluble salt? Why or why not?
-
The following table presents the abundances and masses of the isotopes of zinc. What is the atomic weight of zinc? Isotope 64Zn 66Zn 67 Zn 68Zn 70Zn Abundance 48.6% 27.9% 4.10% 18.8% 0.60% Mass...
-
Pureform, Incorporated, uses the FIFO method of process costing. It manufactures a product passing through two departments. Data for a recent month for the first department follow: Work in process...
-
A local management consulting firm is preparing a business report for the Government of Alberta. In particular, you have been hired by that firm to calculate the weighted average cost of capital (...
-
San Jose Company operates a Manufacturing Division and an Assembly Division. Both divisions are evaluated as profit centers. Assembly buys components from Manufacturing and assembles them for sale....
-
The management of Winstead Corporation is considering the following three investment projects (Ignore income taxes.): Project R $ 84,900 Project S $ 150,500 Investment required Project O $ 46,400...
-
A $ 1 , 0 0 0 face amount 8 % convertible bond has a conversion ratio of 2 0 . The firm s common stock is currently selling at $ 4 0 . If the bond is about to mature, what is its value?
-
What are the steps that organizations take in order to manage information security risks and build a risk matrix? What is involved in each step of this process?
-
A city maintains a solid waste landfill that was 12 percent filled at the end of Year 1 and 26 percent filled at the end of Year 2. During those periods, the government estimated that total closure...
-
Does the sun or the moon have the greater influence in causing tides?
-
A European driving from Paris to Brussels finds she has covered 291 km. How many miles is this?
-
For the circuit in Fig. 5.93 , find v o . 25 k2 100 k2 40 k2 20 k2 20 k2 10 k2 Vo +, 2 V 0+
-
Hannah is a teacher, single, had gross income of $50,000, and incurred the following expenses: Charitable contribution $2,000 Taxes and interest on home 7,000 Legal fees incurred in a tax dispute...
-
The Wilson Company purchased $32,000 of merchanase from the Poole Wholesale Compony. Wisconalso pad $2,500 for treight costs to have the goods thipped to its location The compacy uses the perpetual...
-
If First Interest Bank issues a written note to Joaquin indicating that the bank has received $1,500 as a loan from Joaquin and promises to repay him that amount in the future with interest, this is...

Study smarter with the SolutionInn App


