Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid
Question:
Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid ionization constant for the following ions.
(a) Pyridinium ion
(b) Hydrazinium ion
Table 15.6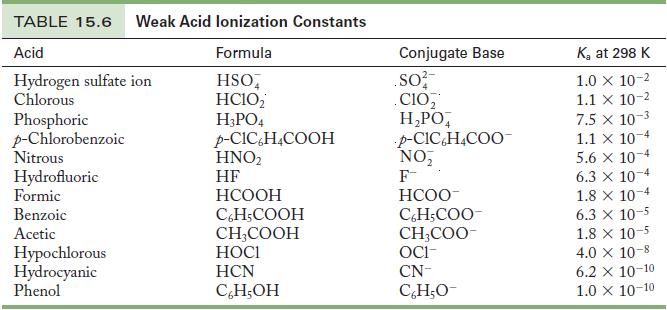
Table 15.8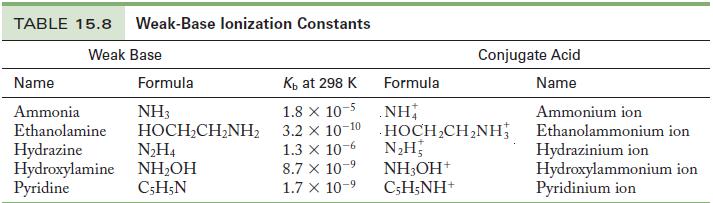
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Answer Acid Ionization Constants of Pyridinium and Hydrazinium Ions a Pyridinium Ion C5H5NH Pyridini...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Is the criterion 6 3CO 2 ) 2 (aq) is found to be 9.11. However, the contribution to the pH from the autoprotolysis of water was ignored. Repeat the calculation of the pH of this solution, taking into...
-
Calcium acetate, Ca(CH 3 CO 2 ) 2 (aq), is used to treat patients with a kidney disease that results in high levels of phosphate ions in the blood. The calcium binds to the phosphates so that they...
-
Although there are extensive tables available for the pK a of weak acids, you might be dealing with an unknown acid or a known acid at an unlisted temperature. You could then use a procedure like...
-
A. What is Alpha One's NOPAT? Why does NOPAT differ from the earnings after taxes? B. Estimate the effective before-tax cost of the long-term debt. c. Estimate the effective after-tax cost of the...
-
Hot exhaust gases are used in a shell-and-tube exchanger to heat 2.5 kg/s of water from 35 to 85C. The gases assumed to have the properties of air, enter at 200C and leave at 93C. The overall heat...
-
Refer to the Applied Psychology in Criminal Justice (Sept. 2009) study of convicted drug dealers' motivations, Exercise 2.102 (p. 77). Recall that the sample of drug dealers had a mean Wanting...
-
For \(a>0\), find the Fourier transform, \(\hat{f}(k)\), of \(f(x)=e^{-a|x|}\).
-
a. Create a network flow model for this problem. (Consider inserting intermediate nodes in your network to assist in meeting the minimum monthly purchase restrictions for each supplier and the...
-
The Stilton Company has the following inventory and creditpurchases during the fiscal year ended December 31, 2023.Beginning619 units @ $78/unitFeb. 10335 units @ $75/unitAug. 2121 2 answers
-
Find the value of K b for the conjugate base of the following organic acids. (a) Picric acid used in the manufacture of explosives; K a = 0.16 (b) Trichloroacetic acid used in the treatment of warts;...
-
Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid ionization constant for the following ions. (a) Hydroxylammonium ion (b) Ammonium ion Table 15.6 Table 15.8
-
The polonium isotope 211 Po is radioactive and undergoes alpha decay. In the decay process, a 211 Po nucleus at rest explodes into an alpha particle (a 4 He nucleus) and a 207 Pb lead nucleus. The...
-
A home builder is looking for the best place to purchase land to build a new home. He researches 10 areas near him to find the number of homes sold there in the past month. He collects the following...
-
(5) For each of the following state whether the code is legal or illegal and say why. (a) int fl(int x); int main() { cout < < fl(5)++; } (b) int &f2(int x); int main() } cout < < f2(5)++; (c) const...
-
Required information In an acoustics experiment, a piano string with a mass of 4.81 g and a length of 69.9 cm is held under tension by running the string over a frictionless pulley and hanging a...
-
The following alphabetized list of selected adjusted account balances is from the records of Jasper Company on December 31, 2023: Accounts Payable Accumulated Depreciation-Equipment Estimated...
-
7. A point charge with a mass of 4.8x10-10 kg is moving with a constant velocity of 3.8x105 m/s to the left. It enters a region with an electric field of 6.1x107 N/C to the right, and after it...
-
1. What was the original function of just war theory with which Walzer agrees? 2. In what sense was the theory an argument of the religious center, according to Walzer, and how did the faculty of the...
-
Prove the formula for (d/dx)(cos-1x) by the same method as for (d/dx)(sin-1x).
-
Given reg [3:0] A = 4'b1101; reg [2:0] B = 3'b111; reg signed [3:0] C = 4'b1101; reg signed [2:0] D = 3'b111; reg signed [7:0] S; Evaluate i. S = A + B; ii. S = A + B + 0; iii. S = C + D; iv. S = C +...
-
Given integer A = 8'shA5; integer B = 8'shB6; integer C = 8'hA5; integer D = 8'hB6; reg signed [31:0] S; Evaluate i. S = A + B; ii. S = A + B + 0; iii. S = C + D; iv. S = C + D + 0; v. S = A + D; vi....
-
A 4-bit magnitude comparator chip (e.g., 74LS85) compares two 4-bit numbers A and B and produces outputs to indicate whether A < B, A = B, or A > B. There are three output signals to indicate each of...
-
The company is considering paying the retail coordinator an incentive commission of $0.10 per pair of earrings. This will be on top of the salespeople's commission and there will be no change in...
-
. TB MC Qu. 16-86 Carmen Company has an asset that cost... Carmen Company has an asset that cost $11,000 and currently has accumulated depreciation of $7,000. Suppose the firm sold the asset for...
-
(LO3) In 2012, Carow sold 3,000 units at $500 each. Variable expenses were $250 per unit, and fixed expenses were $250,000. The same selling price is expected for 2013. Carow is tentatively planning...

Study smarter with the SolutionInn App


