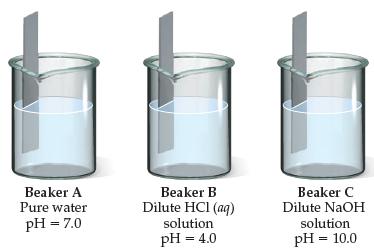
Bars of iron are put into each of the three beakers as shown here. In which beakerA,
Question:
Bars of iron are put into each of the three beakers as shown here. In which beaker—A, B, or C—would you expect the iron to show the most corrosion?
Transcribed Image Text:
Beaker A Pure water Beaker B Dilute HCI (aq) solution Beaker C Dilute NaOH pH = 7.0 solution pH = 4.0 pH = 10.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
The Evolution of the Fashion Industry: Consider the market for clothes and suppose there exist 100 different styles that can be produced and can be arranged (and equally spaced) on a circle. Among...
-
In the U.S., Presidential elections are not won by the candidate who wins the popular vote nationally. (If they were won in this way, Al Gore would have become President in 2000.) Rather, each state...
-
In cases 1-3 below, assume that Division A has a product that can be sold either to Division B of the same company or to outside customers. The managers of both divisions are evaluated based on their...
-
What is GST?
-
What is meant by indirect least squares? When is it used?
-
(Two Differences, One Rate, Beginning Deferred Balance, Compute Pretax Financial Income) Shamess Co. establishes a $90 million liability at the end of 2010 for the estimated litigation settlement for...
-
Solve the general logistic problem, \[\begin{equation*} \frac{d y}{d t}=k y-c y^{2}, \quad y(0)=y_{0} \tag{4.86} \end{equation*}\] using separation of variables. 0.6 0.4 0.2 0 -0.2 -0.4 Nonlinear...
-
Bartlet Financial Services Company holds a large portfolio of debt and stock securities as an investment. The total fair value of the portfolio at December 31, 2010, is greater than total cost. Some...
-
Companies U and L are identical in every respect except that U is unlevered while L has $16 million of 7% bonds outstanding. Assume: (1) All of the MM assumptions are met. (2) Both firms are subject...
-
1. a. Consider 315 grams of original cement, determine the degree of hydration, if anhydrous cement occupies a volume of 50 c.c. 1.b. For w/c=0.63, degree of hydration=1 and 315 g of original cement,...
-
The electrodes in a silver oxide battery are silver oxide (Ag 2 O) and zinc. Which electrode acts as the anode?
-
In each of the following balanced oxidationreduction equations, identify those elements that undergo changes in oxidation number and indicate the magnitude of the change in each case. 2MnO 4 - (aq) +...
-
A thin electronic circuit board, 0.1 m by 0.1 m in size, is to be cooled in air at 25?C. The board is placed in a vertical position and the back side is well insulated. If the heat dissipation is...
-
Question 1 : Why does the preparation of the financial report need adjusting entries? Question 2 : Please use one or two sentences to explain the background of the XBRL mandatory?Question 3 : Please...
-
How do power dynamics manifest within diverse organizational contexts, and what are the implications for employee motivation, satisfaction, and performance ?
-
After a year your costume company has the following balance sheet items at year end. Long - term debt: $ 1 5 , 0 0 0 Cash: $ 5 , 0 0 0 Accounts payable: $ 3 0 , 0 0 0 Long - term assets: $ 2 0 , 0 0...
-
Wilco inc declares a large stock dividned in the current reporting period. How will this tranascation affect the accouting equation
-
Describe how the federal funding of states has changed between AFDC and TANF. What are some of the implications of these changes both over the business cycle and in the long-run. What are the...
-
Currently in the American population of people aged 65 years old and older, there are approximately 130 women for every 100 men. In a nursing home with 280 male residents over the age of 65, how many...
-
What is a manufacturing system?
-
Chlorine is widely used to purify municipal water supplies and to treat swimming pool waters. Suppose that the volume of a particular sample of Cl2 gas is 8.70 L at 895 torr and 24oC. (a) How many...
-
Many gases are shipped in high-pressure containers. Consider a steel tank whose volume is 55.0 gallons that contains O2 gas at a pressure of 16,500 kPa at 23oC. (a) What mass of O2 does the tank...
-
In an experiment reported in the scientific literature, male cockroaches were made to run at different speeds on a miniature treadmill while their oxygen consumption was measured. In one hour the...
-
Use the following assumptions below to answer the following questions. Asking Price: $249,500 Total Sq. Ft.: 2,028 Total Units: 2 Avg. Monthly Rent per Unit: $1,625 Annual Rent Growth: 3.0%...
-
GAAP requires that O all companies that issue an annual report should issue interim financial reports. O the three basic financial statements should be presented each time an interim period is...
-
(a) What is a Pre-Acquisition Elimination Entry and what does it do to the Group Consolidation? b) Why does the Pre-Acquisition Elimination Entry change from the date of acquisition to subsequent...

Study smarter with the SolutionInn App


