Use the solubility-product constant for Cr(OH) 3 (K sp = 6.7 10 -31 ) and the
Question:
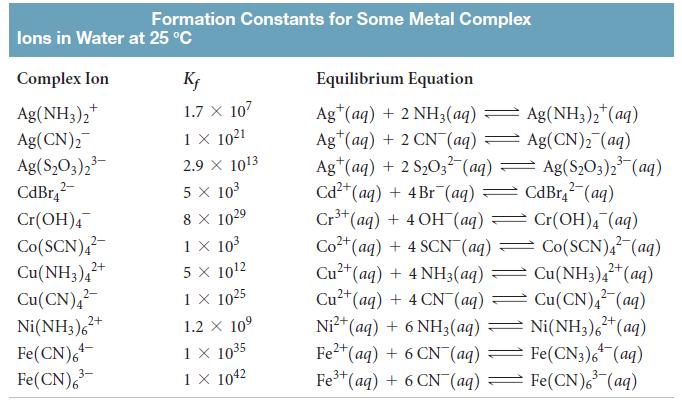
Use the solubility-product constant for Cr(OH)3 (Ksp = 6.7 × 10-31) and the formation constant for Cr(OH)4 from Table 17.1 to determine the concentration of Cr(OH)4 in a solution that is buffered at pH = 10.0 and is in equilibrium with solid Cr(OH)3.
Transcribed Image Text:
Formation Constants for Some Metal Complex lons in Water at 25 °C Complex Ion K; Equilibrium Equation Ag(NH3),* Ag(CN)2 Ag(S,O3),3- CdBr,- Ag" (aq) + 2 NH3(aq) = Ag(NH3)2*(aq) Ag*(aq) + 2 CN (aq) Ag(CN)2 (aq) Ag*(ag) + 2 S203 (aq) = Ag(S,03)2 (aq) Cd2+(aq) + 4Br (aq) Cr*(aq) + 4OH (aq) Cr(OH), (aq) Co2+(aq) + 4 SCN (aq) Cu2+(aq) + 4 NH3(aq) Cu2+(aq) + 4 CN (aq) = 2+(aq)+6 NH3(aq) 1.7 X 107 Ag(NH3),*(aq) 1 × 1021 2.9 x 1013 Ag(S,O3)2 (aq) 5 x 103 CdBr, (aq) Cr(OH)4 8 X 1029 Co(SCN),- Cu(NH3),+ Cu(CN), Ni(NH3),+ Fe(CN),- Fe(CN), 1 x 103 5 x 1012 1X 1025 Co(SCN), (aq) Cu(NH3),*(aq) Cu(CN), (aq) 2+ 2- Ni²* (aq) + 6 NH3(aq) = Ni(NH3),*(aq) Fe"(aq) + 6 CN (aq) = Fe(CN3), (aq) Fe (aq) + 6 CN (aq) 1.2 X 10° 1 X 1035 1 X 1042 4- 3- = Fe(CN), (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
The formation of complex ion CrOH4 involves two reaction steps CrOH3s Cr 3 aq ...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Question Posted:
Students also viewed these Sciences questions
-
Calculate the solubility-product constant for each of the following substances, given that the molar concentrations of their saturated solutions are as indicated: (a) RaSO4 (6.6 x 10-6 M). (b)...
-
The solubility-product constant for Ce(IO3)3 is 3.2 x 10-10. What is the Ce3+ concentration in a solution prepared by mixing 50.00 mL of 0.0450 M Ce3+ with 50.00 mL of? (a) Water? (b) 0.0450 M IO3-?...
-
The solubility-product constant for barium permanganate, Ba(MnO 4 ) 2 , is 2.5 10 -10 . Assume that solid Ba(MnO 4 ) 2 is in equilibrium with a solution of KMnO 4 .What concentration of KMnO 4 is...
-
Linking every transport stakeholder together and ensuring seamless travel across Europe is a dream. With this objective, Amadeus, a leading global travel technology player, initiated a novel idea of...
-
What is wrong with this statement of purpose? PURPOSE: Determine if it takes too long to get cash from the automated teller machine during the lunch hour. Give an improved statement of purpose.
-
A nonnegative integer variable \(y\) that is geometric distributed has density (or more formally probability mass function) \(f(y)=(y+1)(2 \lambda)^{y}(1+2 \lambda)^{-(y+0.5)}, y=\) \(0,1,2, \ldots,...
-
An eight-year, \(6 \%, \$ 1,000\) bond (cash interest payable annually) is sold to yield \(6 \%\) interest. Compute the bond selling price.
-
Proration of overhead. (Z. lqbal, adapted) The Zaf Radiator Company uses a normal-costing system with a single manufacturing overhead cost pool and machine-hours as the cost-allocation base. The...
-
Thank you. You work for a firm whose home currency is the South Korean Won (KRW) and that is considering a foreign investment based in Spain. The investment yields expected after-tax Euro (EUR) cash...
-
Lake Company obtained authorization to issue 10-year bonds with a face value of $5 million. The bonds are dated June 1, 2011, and have a contract rate of interest of 6 percent. They pay interest on...
-
Tooth enamel is composed of hydroxyapatite, whose simplest formula is Ca 5 (PO 4 ) 3 OH, and whose corresponding Ksp = 6.8 10 -27 .As discussed in the Chemistry and Life box on page 730, fluoride in...
-
Calculate the solubility of Mg(OH) 2 in 0.50 M NH 4 Cl.
-
If you deposit %10,000 in a bank account that pays 10 percent interest annually, how much would be in your account after 5 years?
-
(b) In an experiment to investigate niacin retention in vegetables as a function of cooking temperature (A), sieve size (B), type of processing (C), and cooking time (D), each factor was held at two...
-
The field known as "behavioral economics" branched out from traditional neoclassical economics beginning in the 1970s. 1) Explain the theoretical basis for the distinction between traditional...
-
a) In October 2022, Mr. Steve travels to Malaysia and exchanges $200 at the rate of RM4.60/USD1. Based on the information given, answer the following questions. i. In November 2022, the exchange rate...
-
The graph in the figure displays the sample sizes and percentages of people in different age and gender groups who were polled concerning their approval of one mayor's actions in office. The total...
-
A 42-year-old woman has just suffered her 3rd miscarriage. She has no living children. She and her husband have been trying to have children for the last 5 years. Considering your knowledge of...
-
Tracy owns a nondepreciable capital asset held for investment. The asset was purchased for $250,000 six years earlier and is now subject to a $75,000 liability. During the current year, Tracy...
-
Inexhaustible collections of ONPOs are not required to be capitalized or depreciated, if certain criteria are met. Why is this so, and what accounting and reporting recognition, if any, is required...
-
A 50-lossless line of length l = 0 375connects a 300-MHz generator with Vg = 300 V and Zg = 50 to a load ZL. Determine the time-domain current through the load for: (a) ZL =...
-
A generator with Vg = 300 V and Zg = 50 is connected to a load ZL = 75 through a 50-lossless line of length l = 0 15. (a) Compute Zin, the input impedance of the line at the...
-
If the two-antenna configuration shown in Fig. 2-41 (P2.32) is connected to a generator with Vg =250 V and Zg = 50 , how much average power is delivered to each antenna?
-
In the SAP system the Display Document Flow function a. Allows a user to access any of the documents involved in the sales process. b. Shows the completion status of the documents in the sales...
-
To arrive at operating cash flows, you should start with _______, _______ non-cash items and then adding or subtracting changes in working capital. gross profit, subtracting net income, subtracting...
-
Two dimensions within the project management process are a)Technical and sociocultural b)Cost and time c)Planned and unexpected d) Established and new e)Unique and reoccurring

Study smarter with the SolutionInn App


