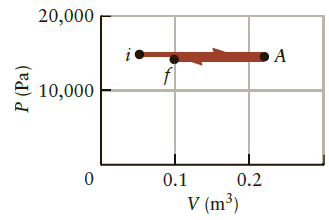
A system undergoes a two-step process as sketched in Figure P16.21. The system first expands at constant
Question:
A system undergoes a two-step process as sketched in Figure P16.21. The system first expands at constant pressure from state I to state A and then it is compressed with the pressure again held fixed, ending with the system in state f. What is the approximate amount of work done by the system during each step?
Figure P16.21
Transcribed Image Text:
20,000 i 10,000 0.1 0.2 V (m³) P (Pa)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
This is an isobaric expansion and compression so the work done in each case is the ...View the full answer

Answered By

Saud Ur Rehman
Evaluating manufacturing processes by designing and conducting research programs; applying knowledge of product design, fabrication, assembly, tooling, and materials; conferring with equipment vendors; soliciting observations from operators. Developing manufacturing processes by studying product requirements; researching, designing, modifying, and testing manufacturing methods and equipment; conferring with equipment vendors. Keeping equipment operational by coordinating maintenance and repair services; following manufacturer's instructions and established procedures; requesting special service.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

College Physics Reasoning and Relationships
ISBN: 978-0840058195
2nd edition
Authors: Nicholas Giordano
Question Posted:
Students also viewed these Sciences questions
-
A system undergoes a two-step process. In the first step, the internal energy of the system increases by 228 J when 166 J of work is done on the system. In the second step, the internal energy of the...
-
A system undergoes a process between two fixed states first in a reversible manner and then in an irreversible manner. For which case is the entropy change greater? Why?
-
A system undergoes a process consisting of the following two steps: Step 1: The system absorbs 72 J of heat while 35 J of work is done on it. Step 2: The system absorbs 35 J of heat while performing...
-
Explain why b x = e x ln b .
-
You notice in the WSJ a bond that is currently selling in the market for $1,070 with a coupon of 11% and a 20-year maturity. Using annual compounding, calculate the promised yield on this bond.
-
Pedro Bourbone is the founder and owner of a highly successful small business and, over the past several years, has accumulated a significant amount of personal wealth. His port-folio of stocks and...
-
A confidence interval for 1 is to be constructed from a sample of 20 points. How many degrees of freedom are there for the critical value?
-
Shiloh Acres sponsors a defined-benefit pension plan. The corporations actuary provides the following information about the plan. The average remaining service life per employee is 10 years....
-
Directions: Evaluate the logarithms. log, 243 log, 343 log 16 4 log 16
-
The uniform bar OC of length L pivots freely about a horizontal axis through O. If the spring of modulus k is unstretched when C is coincident with A, determine the tension T required to hold the bar...
-
You have 3.2 moles of a monatomic ideal gas that are compressed isothermally. (a) If the pressure changes from 4.5 10 5 Pa to 8.5 10 5 Pa and the initial volume was 0.50 m 3 , what is the final...
-
Consider the process described by Figure P16.21. If 500 J of heat are added to the system during the second step, what is the change in the internal energy of the system during this step? Figure...
-
Derive Eqs. (9.105) and (9.106). Equation 9.105 Equation 9.106 SNRDD-PIN rn Prec 2hvRb = (nrno)/2,
-
Identify treatments and experimental units in the following scenarios. a. City A would like to evaluate whether a new employment training program for the unemployed is more effective compared to the...
-
Nowhere is the impact of creative destruction more apparent than on the internet. We can be sure that the growth of the internet will continue to spawn new innovations challenging the survivability...
-
Show that, for irrotational axisymmetric flows in cylindrical coordinates, the streamfunction satisfies the following equation: \[\frac{\partial^{2} \psi}{\partial r^{2}}-\frac{1}{r}...
-
Explain why the sample variance formula is \[\sum_{i=1}^{N} \frac{\left(y_{i}-\bar{y} ight)^{2}}{N-1}\] instead of \[\sum_{i=1}^{N} \frac{\left(y_{i}-\bar{y} ight)^{2}}{N}\]
-
Things looked good for Mylan NV, a leading generic pharmaceutical drug maker, midday on November 13, 2015, as the early results of the firms campaign to acquire Perrigo appeared promising. The votes...
-
A thin layer of liquid, draining from an inclined plane, has a velocity profile v x v 0 (2y/h - y 2 /h 2 ), where v 0 is the surface velocity. If the plane has width 10 cm into the paper, determine...
-
Provide examples of a situations in which environmental disruptions affected consumer attitudes and buying behaviors.
-
Write the node-voltage equations by inspection and then determine values of V 1 and V 2 in the circuit of Fig. 3.114 . 41x V2 V, 7A 5S 1S 2 S 20 A
-
For the circuit shown in Fig. 3.113 , write the nodevoltage equations by inspection. 20 k2 50 mA 10 k2 v2 5 k2 V3 10 k2 40 mA 100 mA 5 k2
-
Using Fig. 3.112 , design a problem, to solve for V o , to help other students better understand nodal analysis. Try your best to come up with values to make the calculations easier. Find the voltage...
-
Detail the impact of personality and behavior on "organizational fit" Explain how personality has an impact on organizational fit or team cohesion. Explain how behavior has an impact on...
-
What is the context for risk management? Question 2: Why is it important to set the scope for risk management? Question 3: Explain why it is necessary for a business to document the identified risks...
-
List and explain four reasons you might consider refurbishing an existing piece of fire apparatus compared to replacement. What are the considerations you have to think of to compare refurbishment...

Study smarter with the SolutionInn App


