Figure P16.23 shows a system that is compressed from an initial state i to a final state
Question:
Figure P16.23 shows a system that is compressed from an initial state i to a final state f. If the internal energy of the system decreases by 5000 J, approximately how much heat was added to the system?
Figure P16.23
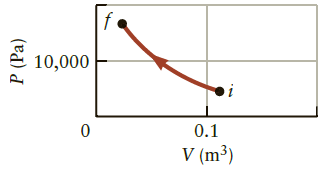
Transcribed Image Text:
10,000 0.1 V (m³) P (Pa)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (20 reviews)
The heat transferred internal energy and work done on the system are related by the first law of the...View the full answer

Answered By

Sarfraz gull
have strong entrepreneurial and analytical skills which ensure quality tutoring and mentoring in your international business and management disciplines. Over last 3 years, I have expertise in the areas of Financial Planning, Business Management, Accounting, Finance, Corporate Finance, International Business, Human Resource Management, Entrepreneurship, Marketing, E-commerce, Social Media Marketing, and Supply Chain Management.
Over the years, I have been working as a business tutor and mentor for more than 3 years. Apart from tutoring online I have rich experience of working in multinational. I have worked on business management to project management.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

College Physics Reasoning and Relationships
ISBN: 978-0840058195
2nd edition
Authors: Nicholas Giordano
Question Posted:
Students also viewed these Sciences questions
-
A gas is compressed from an initial volume of 0.42 m3 to a final volume of 0.12 m3. During the quasi-equilibrium process, the pressure changes with volume according to the relation P + aV + b, where...
-
A gas is compressed from an initial volume of 0.42 m3 to a final volume of 0.12 m3. During the quasi-equilibrium process, the pressure changes with volume according to the relation P = aV + b, where...
-
Hydrogen sulfide gas is compressed from an initial state of 400 K and 5 bar to a final state of 600 K and 25 bar. Estimate (H and (S?
-
Let Find x , y , xy , and yx , and state the domain for each partial derivative. f(x, y) = [y, y = 0 1-y, y < 0.
-
The chart shows the number of global corporate bond issues for which Standard & Poor's issued ratings upgrades or downgrades every year from 1981 to 2014. a. What is the trend in the number of...
-
Carl and Carol have salaries of $14,000 and $22,000, respectively. Their itemized deductions total $8,500. They are married and both are under age 65. a. Compute their taxable income assuming they...
-
The following table lists values measured for 60 consecutive eruptions of the geyser Old Faithful in Yellowstone National Park. They are the duration of the eruption (x1), the duration of the dormant...
-
Rob Company purchases a 90% interest in Venus Company for $418,500 on January 1, 2013. Any excess of cost over book value is attributed to equipment, which is being depreciated over 20 years. Both...
-
How much money do you need to live comfortably each year? Everyone could have different answer here. So, let's make it 1million here, and this will be your annual income at retirement with this same...
-
Draw the supply and demand curves for frozen yogurt. Show equilibrium. 2. Show and explain what will be the first thing that will happen in the market for frozen yogurt when each of the following...
-
Consider the process described by Figure P16.21. If 500 J of heat are added to the system during the second step, what is the change in the internal energy of the system during this step? Figure...
-
An ideal monatomic gas expands adiabatically from an initial temperature of 400 K and volume of 4.5 m 3 to a final volume of 12 m 3 . If the initial pressure is 1.5 P atm , how much work is done on...
-
Find parametric equations and symmetric equations for the line. The line through (-6, 2, 3) and parallel to the line 1/2 x = 1/3 y = z + 1
-
What are counterbiasing questions and what are they used for?
-
Do health care leaders have a responsibility to be culturally competent? Why or why not?
-
What is a split-ballot technique and what is it used for?
-
Actions required to gain regulatory approval usually involve selling off some portion of the combined business in countries where increased concentration is a concern. In doing so, the economics of...
-
Why do health care leaders have a higher need for ethical behavior than might be expected in other settings?
-
Water flows steadily through the piping junction, entering section 1 at 0.0013 m 3 /s. The average velocity at section 2 is 2.1 m/s. A portion of the flow is diverted through the shower head, which...
-
The electric field due to a line charge is given by where l is a constant. Show that E is solenoidal. Show that it is also conservative. E =
-
Given the circuit in Fig. 3.53 , calculate the currents i 1 through i 4 . i3 40 10 40 20
-
Obtain v a in the circuit of Fig. 3.54 . 120 V +- 60 V 120 k2 120 k2 Vo 30 k2
-
Determine v 1 and v 2 in the circuit of Fig. 3.101 . 20 10 10 100 V 10 10 20
-
Down Under Products' sales budget for the next four months is as follows: Unit Sales April May June July 82,000 90,000 122,000 96,000 The company wants its ending inventory levels to equal 15% of the...
-
For a cyclotron, apply the relationship between the particle's mass and charge, the magnetic field, and the frequency of circling. Suppose a cyclotron is operated at B = 1.57 T and has a dee radius R...
-
Read the linked article re: increasing prices, and review the chart as well. Comment on where you think prices (in general) are headed and why?...

Study smarter with the SolutionInn App


