A manometer, containing water; is used to measure the pressure of a gas supply in a school
Question:
A manometer, containing water; is used to measure the pressure of a gas supply in a school laboratory. Its reading is h cm of water.
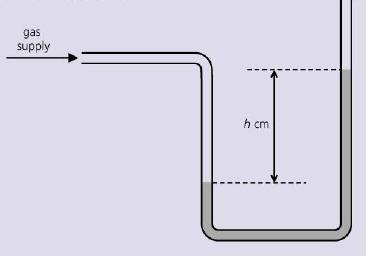
Why is it better to use water in the manometer, rather than mercury?
A. With mercury, a narrower tube would be needed.
B. With mercury, a wider tube would be needed.
C. With mercury, it would be too large.
D. With mercury, h would be too small.
Transcribed Image Text:
gas supply h cm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
B With mercury a wider tube would be needed Water has a higher densi...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A differential manometer is used to measure the pressure change caused by a flow constriction in a piping system as shown. Determine the pressure difference between points A and B in psi. Which...
-
A U-tube mercury manometer is used to measure the pressure at a point on the wing of a wind-tunnel model. One side of the manometer is connected to the model, and the other side is open to the...
-
A thermocouple (0.8-mm-OD wire) is used to measure the temperature of quiescent gas in a furnace. The thermocouple reading is 165?C. It is known, however, that the rate of radiant heat flow per meter...
-
Solve each equation. a. x = x b. x(x 8) = 4(x - 9) -
-
Suppose that a BF3 molecule approaches the lone pair on the N atom of an :NH3 molecule, and that a bond forms between the B atom and the N atom. Consider the arrangement of electron pairs about the B...
-
Suppose an investigator takes a random sample of \(n=50\) birth weights from several teaching hospitals located in an inner-city neighborhood. In her random sample, the sample mean \(\bar{x}\) is...
-
Match threats in the first column to appropriate control procedures in the second column. Threat 1. Sending incorrect merchandise Control Procedure a. Perform a completeness check 2. Crediting...
-
The Colson Company issued $311,000 of 10% bonds on January 1, 2011. The bonds are due January 1, 2016, with interest payable each July 1 and January 1. The bonds are issued at 98. Prepare the journal...
-
Basic Modern Road Bike Component Identification Guide 1. Brake Levers (CHINA)1.50 2. Cassette (CHINA) 3.50 3. Chain (CHINA) 2.25 4. Chain Stays (CHINA)2.50 5. Chainrings (CHINA) 3.50 6. Crank arm...
-
Ouellette Co. adjusts its books monthly. On June 30, 2014, notes receivable include the following: Interest is payable on the first day of each month for notes with terms of one year or longer....
-
Some fat purchased from a shop is supplied as the block shown below. Use the information in the diagram to calculate a. The volume of the block (in cm 3 ), b. The density of the fat. Give your answer...
-
This question is about trucks from a small train in a park: One truck, mass 240 kg, is given a push and released at a speed of 3 m/s. It collides with a bigger, stationary truck of mass 360 kg and...
-
General Telephone Company of Florida owned more than 1 percent of the stock of Florida Telephone Corporation. General sought to examine the latters stock records in order to make a list of the names,...
-
12) What is the output of the following code? (5points) x=[] def F(y,x): L=0 for i in range(1,7): L-=1 if y[i]
-
Assume that you are the leadership team of a convenience store chain that has more than 300 outlets. The company is facing an inventory shrinkage problem, and store managers report that the main...
-
Gamers Technology ( GT ) is a private company with a December 3 1 st fiscal year end. GT is an online magazine which it sells on a 1 2 - month subscription basis. To meet the daily demands of its...
-
You, CPA, work for Mercer Consulting Ltd. It is now February 12, 2019, and your boss, Patricia Mercer, has just returned from a meeting with Adam Burke of Burke Entertainment Ltd. (BEL). Patricia...
-
function addChar ( input , character ) { if ( input . value = = null | | input.value = = " 0 " ) input.value = character else input.value + = character } function cos ( form ) { form.display.value =...
-
Fastest Company has a debt rating of A and a tax rate of 35 percent. The current long-term government bond yield is 2 percent. Suppose the typical spread between long-term government yields and...
-
Evaluate each logarithm to four decimal places. log 0.257
-
Assign an IUPAC name for each of the following compounds: a. b. c. d. ,
-
The 1 H NMR spectrum of a compound with molecular formula C 7 H 14 O 3 exhibits only three signals, and all three signals appear above 2 ppm (downfield of 2 ppm) on the spectrum. Propose a structure...
-
How would you use 1 H NMR spectroscopy to distinguish between the following compounds? (a) (b) (c) (d) (e) (f) . . . CI
-
The following data come from the financial records of Adams Corporation for Year 3. Sales $845,000 Interest expense 5,200 Income tax expense 27,500 Net income 24,000 Required How many times was...
-
The Better Future Inc. stock is selling for $32.60 a share based on a 14 percent rate of return. What is the amount of the next annual dividend if the dividends are increasing by 3.5 percent...
-
Fill out a Lease Abstract: NDUSTRIAL BUILDING LEASE THIS LEASE is made as of this Z 1 day of June, 2 0 1 0 betwe PROPERTIES TRUST, a Marland real estate investment trust ( " Landlord " ) , and INC.,...

Study smarter with the SolutionInn App


