The diagram below shows the mass of measuring cylinder before some liquid is poured into it and
Question:
The diagram below shows the mass of measuring cylinder before some liquid is poured into it and then after.
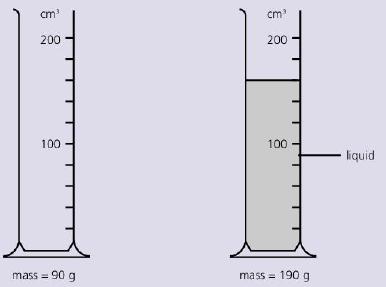
What is the density of the liquid?
A. 100/160 g/cm3
B. 100/130 g/cm3
C. 190/160 g/cm3
D. 100/130 g/cm3
Transcribed Image Text:
cm cm 200 200 100 100 liquid mass = 90 g mass = 190 g !!
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Option A is Correct answer Density Mass Volume ...View the full answer

Answered By

Muhammad Waqas Hanif
I have experience of 2 years teaching. Also I have sound knowledge of research. I teach students at High School Level to Master Level.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The diagram below shows the various short-run cost curves for a perfectly competitive firm. a. Based on the diagram, and the assumption that the firm is maximizing its profit, fill in the table. The...
-
The diagram below shows the Canadian market for leather shoes, which we assume to be competitive. The world price is pw. If the Canadian government imposes a tariff of t dollars per unit, the...
-
The diagram below shows the short run demand curve (D), marginal revenue curve (MR), average total cost curve (ATC), and marginal cost curve (MC) for a firm in a monopolistically competitive market....
-
Suppose the given numbers for a mental calculation (see Problem 59) are 10x + y and 10x + z. Notice that these two numbers have the same tens digit. Also assume that y + z = 10, which says that the...
-
Use the VSEPR model to predict the geometry of the following ions: a. N3 b. BH4 c. SO32 d. NO2
-
Below are amounts (in millions) reported in each of these companies annual reports? Required: For each company, calculate the receivables turnover ratio and the average collection period. Which...
-
According to the Organizational Culture Inventory (OCI), an organization in which members are encouraged to work together in ways that meet higher-order human needs is classified as having a...
-
The Fanta Company presents you with the following account balances taken from its December 31, 2007 adjusted trial balance: Inventory, January 1, 2007 ............. $ 43,000 Selling expenses...
-
Follow the instructions below. (a) Rewrite the decimal in the sentence below as a percentage. The value had decreased to 0.067 of the purchase price. (b) Rewrite the percentage in the sentence below...
-
The Prompt Corporation purchases product from two suppliers, A and B. Supplier A is located in Austin, TX, and supplier B in Carthage, TN. Supplier A supplies component a and supplier B supplies...
-
Isotopes of the radioactive element uranium occur naturally in small proportions in some rocks. The table gives information about one uranium isotope. a. How many neutrons are there in an atom of...
-
Drops of water from a cracked gutter fall past the window of an IGCSE Physics students room, as shows in the diagram. The student uses a digital stopwatch to find the time between one drop and the...
-
A cyclotron produces a 3.50-A proton beam of 60-MeV energy. The protons impinge and come to rest inside a 50-g copper target within the vacuum chamber. (a) Determine the number of protons that strike...
-
How to prepare a deposition summary in different ways?
-
An auditor uses capital letters and Arabic numbers to index her working papers. She decides to insert two worksheets between worksheets C-5 and C-6, and one worksheet between D-8 and D-9. How should...
-
Internal auditors should be concerned with improving conditions but they should not demand perfection. Why?
-
Who can attend a deposition?
-
What benefits accrue from reporting deficiency findings currently to management?
-
How can information itself provide a competitive advantage to an organization? Give two examples. For each example, describe its associated risks.
-
For a nonzero constant a, find the intercepts of the graph of (x 2 + y 2 ) 2 = a 2 (x 2 - y 2 ). Then test for symmetry with respect to the x-axis, the y-axis, and the origin.
-
While 13 C is the main contributor to the (M+1) + peak, there are many other elements that can also contribute to the (M+1) + peak. For example, there are two naturally occurring isotopes of...
-
Below are mass spectra for four different compounds. Identify whether each of these compounds contains a bromine atom, a chlorine atom, or neither. a. b. c. d. 100- 80- 60- 60- 40 20- 0- 60 70 10 20...
-
For each of the following molecules, determine the number of carbon atoms present, and then determine the number of hydrogen atoms connected to each carbon atom: a. b. c. d. e. f. g. h. i. j. k. l....
-
The consolidated financial statements can be prepared by using which procedures? Question 1Select one: a. Pro forma consolidation journal entries prepared from the analysis of owners' equity and then...
-
Harrison Rentals Company faced the following situations. (Click the icon to view the situations.) Requirement 1. Journalize the adjusting entry needed at December 31, 2023, for each situation....
-
Emma holds a $5,000 portfolio that consists of four stocks. Her investment in each stock, as well as each stock's beta, is listed in the following table: Stock Investment Beta Standard Deviation Omni...

Study smarter with the SolutionInn App


