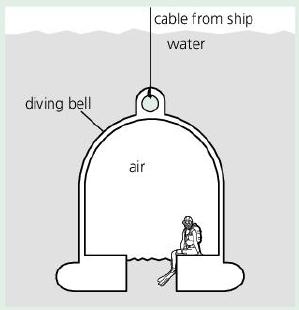
The diving bell below contains trapped air at the same pressure as the water outside. At the
Question:
The diving bell below contains trapped air at the same pressure as the water outside. At the surface, air pressure is 100 kPa. As the bell descends, the pressure on it increases by 100 kPa for every 10 m of depth.
a. What is the pressure on the diver at depths of 0 m, 10m, 20m and 30m?
b. At the surface, the bell holds 6 m3 of air. If the bell is lowered to a depth of 20 m, and no more air is pumped into it, what will be the volume of the trapped air? (Assume no change in temperature.)
cable from ship water diving bell air
Step by Step Answer:
aPressure at 0m depth no water only air pressureSo pressure is 100k...View the full answer



Related Video
For this experiment we need-One glass bottle (opening slightly smaller than an egg)-A matchbox-One peeled boiled eggIn this demonstration, the lit matchsticks heat the air inside the bottle. When air is heated it expands and some of it escapes out of the bottle. When the matches go out, the air inside the bottle cools and contracts thus creating a lower air pressure area inside the bottle than outside. Normally, the high-pressure air outside the bottle would come rushing in to equalize the low-pressure air in the bottle. The problem is that the egg is in the way. The air molecules on the outside of the bottle push the egg into the bottle.•Procedure• Place a glass bottle on the table.• Boil a normal-sized egg and• Place the boiled egg on the face or neck of the bottle.• We can see the egg cannot be pushed into the bottle because of air inside the bottle.• Now light the matches at the same time and drop them into the bottle.• Once the matchsticks have been dropped into the bottle Quickly Place the egg over the neck of the bottle.• You can see the egg is sucked into the bottle.This represents the air pressure and characteristics of air.
Students also viewed these Sciences questions
-
A 3 m3 tank initially contains air at 100 kPa and 25oC. The tank is connected to a supply line at 550 kPa and 25oC. The valve is opened, and air is allowed to enter the tank until the pressure in the...
-
A 10 m3 rigid tank contains air at 200 kPa and 150oC. A 1 kW internal heater is turned on. Determine the rate of change of (a) Stored energy (b) Temperature. (c) Pressure of air in the tank. Use the...
-
Air is pumped into and withdrawn from a 10 m3 rigid tank as shown in the accompanying figure. The inlet and exit conditions are as follow. Inlet: v1 = 2 m3/kg, V1 = 10 m/s, A1 = 0.01 m2; Exit: v2 = 5...
-
What are the energies of the two longest-wavelength lines in the Paschen series for hydrogen? What are the corresponding wavelengths? Give your answers to two significant figures.
-
An electric heater coil provided heat to a 15.5-g sample of iodine, I2, at the rate of 3.48 J/s. It took 4.54 min from the time the iodine began to melt until the iodine was completely melted. What...
-
During the period, Sanchez Company sold some excess equipment at a loss. The following information was collected from the companys accounting records: No new equipment was bought during the period....
-
Chesterfield Wanderers is a professional football club that has enjoyed considerable success in recent years. As a result, the club has accumulated 10 million to spend on its further development. The...
-
In 2010, Grand Falls Bank (GFB) had $4,000,000 in business loans at an average interest rate of 3.5 percent as well as $3,200,000 in consumer loans with an average rate of 8 percent. GFB also has...
-
QUESTION 5*2 POINTS Find the equation of the line with slope (1)/(5) and y-intercept (0,1).
-
Following are some of the countys governmental funds. The county has asked you to determine is any of the funds listed should be classified as major funds based on the GASB size criteria. Clearly...
-
A car has a mass of 900 kg. It accelerates from rest at a rate of 1.2 m/s 2 . a. Calculate the time taken to reach a velocity of 30 m/s. b. Calculate the force required to accelerate the car at a...
-
The diagram below shows a uniform metre rule, weight W, pivoted at the 75 cm mark and balanced by a force of 2 N acting at the 95 cm mark. a. Calculate the moment of the 2 N force about the pivot. b....
-
Suppose you wish to estimate a population mean based on a random sample of n observations, and prior experience suggests that = 12.7. If you wish to estimate correct to within 1.6, with probability...
-
Many top-level managers do not understand CIM. Only a small percentage of conversions to CIM began with top-level managers. The push for CIM usually begins with shop-floor engineers. What are the...
-
If df > 90, the distribution is _____________. If df = 15, the distribution is ________________.
-
A hospital administrator has just read a book about JIT. She feels that JIT is a good idea for manufacturing companies, but doesnt think that JIT would be of much use to a service organization....
-
What are the advantages and disadvantages of permitting counter-claims for insurance claims to be offset in financial reports? Consider the views of management, shareholders and a non-governmental...
-
Claire Hunter plans on opening Claire Hunter Floral Designs. She is considering the various types of business organizations and wishes to organize her business with unlimited life and limited...
-
Write an essay in which compare the Christian worldview to the secular worldview of accounting. In this paper, research a topic(s) of choice that will analyze the Christian worldview as compared to...
-
What are the typical record-at-a-time operations for accessing a file? Which of these depend on the current file record?
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (g) (h) SH
-
Paclitaxel (marketed under the trade name TaxolTM) is found in the bark of the Pacific yew tree, Taxus berevifolia, and is used in the treatment of cancer: (a) Draw the enantiomer of paclitaxel. (b)...
-
Predict the major product(s) for each of the following reactions: a. b. c. d. (PPH3)3RHCI H;o*
-
Dr. Minn is selling her physical therapy practice after owning the practice for 20 years. Dr. Linn is going to buy the radiology practice at a higher price because Dr. Minn has over 500 patients and...
-
Dr. Tier owns a non-profit agency in the local community that provides food, shelter, and counseling for disadvantaged youth and families. At the end of the year, the non-profit agency's difference...
-
Comparative financial statements for Weller Corporation, a merchandisi ompany, for the year ending December 31 appear below. The company did not issue any new common stock during the year. A total of...

Study smarter with the SolutionInn App


