A rocket propellant manufacturer is studying the burning rate of propellant from three production processes. Four batches
Question:
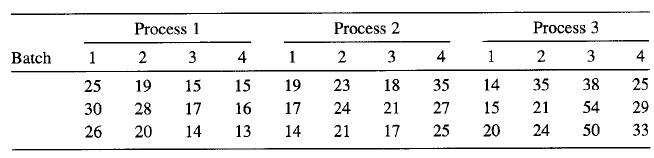
A rocket propellant manufacturer is studying the burning rate of propellant from three production processes. Four batches of propellant are randomly selected from the output of each process and three determinations of burning rate are made on each batch. The results follow. Analyze the data and draw conclusions.
Transcribed Image Text:
Process 1 Process 2 Process 3 Batch 1 3 4 1 2 3 4 1 3 4 25 19 15 15 19 23 18 35 14 35 38 25 30 28 17 16 17 24 21 27 15 21 54 29 26 20 14 13 14 21 17 25 20 24 50 33
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
So to get the GPE of an object you need a formula the formula is MASS X GRAVITY X HEIGHT M X G X H F...View the full answer

Answered By

Hardik Dudhat
I am semi-qualified Chemical Engineering ,I have scored centum in accounting in my senior secondary and in my graduation. I have always helped my fellow students with their concerns on the subject, i have tutored on various tutoring sites in the past and also have taken home tuitions for degree and MBA students. As a tutor, I don't want my students to just get a solution, I want them to understand the concept and never have a doubt in that area thereon and i believe in excelling and not in educating.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The following requirements are given for a solid propellant rocket motor: Sea level thrust: 740 lbf average Duration: 42 sec. Chamber Pressure: 750 psia Operating Temp.: 70 F Propellant: Ammonium...
-
Consider a rocket engine burning hydrogen and oxygen. The total mass flow of the propellant plus oxidizer into the combustion chamber is 287.2 kg/s. The combustion chamber temperature is 3600 K....
-
Two distinct solid fuel propellants, type A and type B, are being considered in a space program activity. Burning rates on the propellant are crucial. Random samples of 20 specimens of the two...
-
5.8 Calculate the U value for the following double- glazed windows assuming the temperatures and the heat transfer coefficients as given in Example 5.1: (a) Ordinary glass with vacuum between the...
-
Suppose that fraction used = 0.5 / 1.0 + Mt for some parameter . Write the discrete-time dynamical system and solve for the equilibrium. Sketch a graph of the equilibrium as a function of . What...
-
How far from a -7.20-C point charge must a +2.30-C point charge be placed for the electric potential energy U of the pair of charges to be -0.400 J? (Take U to be zero when the charges have infinite...
-
What are the steps in developing a personal investment plan?
-
Depreciation ComputationReplacement, Nonmonetary Exchange Goldman Corporation bought a machine on June 1, 2008, for $31,800, f.o.b. the place of manufacture. Freight to the point where it was set up...
-
Pete's group insurance policy specifies that he pays 45?% of expenses associated with orthodontic treatment for his children. If Pete incurs expenses of $4,100?, how much would he? owe? Pete ...
-
Robertos Honey Farm in Chile makes five types of honey: cream, filtered, pasteurized, mlange (a mixture of several types), and strained, which are sold in 1 kilogram or 0.5 kilogram glass containers,...
-
Rework Problem 12-32 using the modified large-sample method described in Section 12-7.2. Compare this confidence interval with the one obtained previously and discuss. Problem 12-32 Consider the...
-
The surface finish of metal parts made on four machines is being studied. An experiment is conducted in which each machine is run by three different operators and two specimens from each operator are...
-
1. What does the restaurant scenario presented above have to do with contingency leadership? 2. In what way might Jessica and the supervisors modify their leadership styles to adapt to the...
-
A company purchased equipment for 2 4 , 0 0 0 what account would be debited in the adjusting entry at the end of the accounting period?
-
First find f' and then find f. f"(x) = 7x3+7x2 - 8x - 6, f'(1) = 4, f(1) = 8. f'(x) = f(x) =
-
A rescue plane flies horizontally at a constant speed searching for a disabled boat. When the plane is directly above the boat, the boat's crew blows a loud horn. By the time the plane's sound...
-
A store like Dollar General strives to offer a little bit of everything. They may not have every brand or flavor of every product type, but they generally have at least one choice for each product...
-
16. A 0.40 m rod has non-uniform linear mass density given by (r) = Ar, where 'r' is the distance from the end of the rod to a point on the rod and the coefficient A has a numerical value of 2.00....
-
It is false that no incendiary devices are contraptions that misfire. Therefore, some incendiary devices are not contraptions that misfire. Use the modified Venn diagram technique to determine if the...
-
Chris Zulliger was a chef at the Plaza Restaurant in the Snowbird Ski Resort in Utah. The restaurant is located at the base of a mountain. As a chef for the Plaza, Zulliger was instructed by his...
-
The following equation describes a certain dilution process, where y(t) is the concentration of salt in a tank of freshwater to which salt brine is being added. Suppose that y(0) = 0. Plot y(t) for 0...
-
The following equation describes the motion of a certain mass connected to a spring, with viscous friction on the surface where f(t) is an applied force. Suppose that f (t) = 0 for t + 0 and f (t) =...
-
The following equation describes the motion of a certain mass connected to a spring, with viscous friction on the surface where f(t) is an applied force. Suppose that f(t) = 0 for t + 0 and f(t) = 10...
-
Chapter 5 Question 11 The market price of a $970000, ten-year, 14% (pays interest semiannually) bond issue sold to yield an effective rate of 12% is $1089568. $1800156. $1079588. $1081254.
-
Chapter 11: Management of transaction exposure (pages 423 and 424) - case description Blades plc has recently decided to expand its international trade relationship by exporting to the United States....
-
FIN 301, Section 002: CORF X McGraw-Hill Education Car X Question 1 - Chapter 8 Qu X Welcome - COMM100 Nev x Course Hero G sams club willow grove ga + C...

Study smarter with the SolutionInn App


