The yield of a chemical process was measured using five batches of raw material, five acid concentrations,
Question:
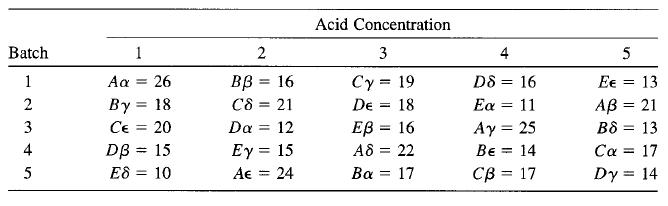
The yield of a chemical process was measured using five batches of raw material, five acid concentrations, five standing times (A, B, C, D, E), and five catalyst concentrations (α, β, γ, δ, ∈). The Graeco-Latin square that follows was used. Analyze the data from this experiment (use α = 0.05) and draw conclusions.
Transcribed Image Text:
Acid Concentration Batch 1 2 4 5 D8 = 16 Εα 11 Ay = 25 1 26 вв 3 16 Cy = 19 Aa Ee = 13 %3D By = 18 Ce = 20 DB = 15 E8 = 10 2 C8 21 De 18 AB = 21 %3D %3D 3 Da = 12 ЕВ 3 16 B8 = 13 %3D Ey = 15 Ae = 24 4 A8 = 22 Be = 14 Ca = 17 %3D %3D 5 Ba = 17 Св 3 17 Dy = 14 %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Batch Acid Time Catalist Yield 1 1 A a 26 2 1 B c 18 3 ...View the full answer

Answered By

Anyango Anyango
Immediately after I finished secondary school waiting to join university I was practising as a tutor of business because of my passion and interests. I have done that for the longest time even though pursuing a different carrier.
I also love playing with figures to solve mathematical problems.
Above all this, I love assisting learners to scale higher and get good grades. Most of the time I would volunteer to teach students in the neighbourhood with their assignments.So far I have seen most of them improve well in their academics and even pass with flying colours and able to join the university.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The yield of a chemical process is being studied. From previous experience yield is known to be normally distributed and = 3. The past five days of plant operation have resulted in the following...
-
A production supervisor at a major chemical company wishes to determine whether a new catalyst, catalyst XA-100, increases the mean hourly yield of a chemical process beyond the current mean hourly...
-
Referring to Exercise 8.21 (page 307), regard the sample of five trial runs (which has standard deviation 19.65) as a preliminary sample. Determine the number of trial runs of the chemical process...
-
93) Clay of the Land is a manufacturer of glazed clay pots. Currently, it produces 300 clay pots per month which it sells through nurseries at a constant price of $5 per pot. Current demand for clay...
-
Find the equilibria of the following discrete-time dynamical system from the graphs of their updating functions Label the coordinates of the equilibria. 10 Time th)
-
Two pulses are moving in opposite directions at 1.0 cm/s on a taut string, as shown in Fig. IS.34. Each square is 1.0 cm. Sketch the shape of the string at the end of (a) 6.0 s; (b) 7.0 s; (c) 8.0 s.
-
The following information for Great Oaks Furniture, a retail furniture and design firm, relates to Exercises 6 through 11. Perform a vertical analysis of the balance sheets and income statements for...
-
The Bartram-Pulley Company (BPC) must decide between two mutually exclusive investment projects. Each project costs $6,750 and has an expected life of 3 years. Annual net cash flows from each project...
-
you were hired as a consultant to Giambono company whose target capital structure is 40% debt, 15% preferred and 45% common equity the after tax cost of debt is 6.00^ the cost of preferred is 7.50%...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Suppose that in Problem 4-14 the data taken on day 5 were incorrectly analyzed and had to be discarded. Develop an appropriate analysis for the remaining data. Problem 4-14 A chemist wishes to test...
-
Suppose that in Problem 4-15 the engineer suspects that the workplaces used by the four operators may represent an additional source of variation. A fourth factor, workplace (, , , ) may be...
-
One mole of ideal gas is confined in a piston-cylinder device, which is 1 foot in diameter. The piston can be assumed weightless and frictionless. The internal and external pressures are both...
-
Fulton Electronics sells televisions and other electronics. All the electronics come with a default one - year warranty against defects. Warranty estimates are recorded every month. Fulton estimates...
-
Dean and sumit have been in partnership for some years running a manufacturing business. Dean receivea a salary of R70 000 a year from the partnership. Interest on salary paid at the rate of 10% and...
-
1. Four identical wooden blocks, initially at rest, are each released from the same height above the ground in all four cases in the diagram below labeled 1-4. Note the description of the friction...
-
Our new business venture of creating a comfortable, luxurious, and modern office spaces accessible for the average person.A direct competitor would be WeWork. Discuss how they are a direct...
-
Segmentation is key to identifying the right target markets. As a start-up company, SOCIAL LITE Vodka knew it had to be extremely focused on segmentation. Its alcoholic beverages were made with 100...
-
Consider the following fictional URL: www.acme.com/products/info.html. What does each part of this URL (separated by slashes) specify?
-
Fill in each blank so that the resulting statement is true. A solution to a system of linear equations in two variables is an ordered pair that__________ .
-
A 20.0-mL solution of 0.005 00 M Sn 2+ in 1 M HCl was titrated with 0.020 0 M Ce 4+ to give Sn 4+ and Ce 3+ . Calculate the potential (versus S.C.E.) at the following volumes of Ce 4+ : 0.100, 1.00,...
-
Would indigo tetrasulfonate be a suitable redox indicator for the titration of Fe(CN) 6 4- with Tl 3+ in 1 M HCl? The potential at the equivalence point must be between the potentials for each redox...
-
Compute the titration curve for Demonstration 15-1, in which 400.0 mL of 3.75 mM Fe 2+ are titrated with 20.0 mM MnO 4 - at a fixed pH of 0.00 in 1 M H 2 SO 4 . Calculate the potential versus S.C.E....
-
Analyze the balance sheet for DEF Inc. as of December 31, 2023, to evaluate its liquidity and financial risk. DEF Inc. Balance Sheet As of December 31, 2023 Assets Current Assets Cash $30,000...
-
Samuel plans to deposit $350 at the beginning of every month into a bank account. The bank account has an interest rate of 6% per year. How much will Samuel have in the account at the end of 2 years?...
-
The Assembly Department of ByteSize, Inc., manufacturer of computers, incurred $250,000 in direct material costs and $90,000 in conversion costs. The equivalent units of production for direct...

Study smarter with the SolutionInn App


