A mixture that contains 40% by mole n-heptane in n-decane is to be separated in a series
Question:
A mixture that contains 40% by mole n-heptane in n-decane is to be separated in a series of flush separators until a stream is obtained that contains at least 95% n-heptane. Determine the number of separators needed, their temperature, and the recovery of n-heptane if all separators are at 1.013 bar and V/L = 3 in all separators. Txy data are given below:
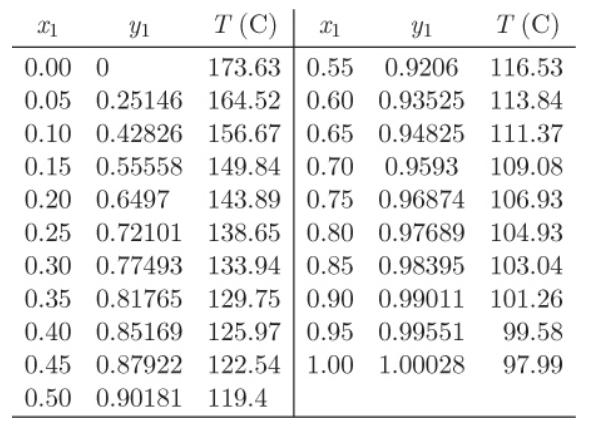
Transcribed Image Text:
X1 T (C) X1 Yı T (C) 0.00 0 173.63 0.55 0.9206 116.53 0.05 0.25146 164.52 0.60 0.93525 113.84 0.10 0.42826 156.67 0.65 0.94825 111.37 0.15 0.55558 149.84 0.70 0.9593 109.08 0.20 0.6497 143.89 0.75 0.96874 106.93 0.25 0.72101 138.65 0.80 0.97689 104.93 0.30 0.77493 133.94 0.85 0.98395 0.35 0.81765 129.75 0.90 0.99011 0.40 0.85169 125.97 0.95 0.99551 0.45 0.87922 122.54 1.00 103.04 101.26 1.00028 0.50 0.90181 119.4 Y1 99.58 97.99
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
To determine the number of separators needed their temperature and the recovery of nheptane in each separator we can use the concept of flash distilla...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9780132693066
1st Edition
Authors: Themis Matsoukas
Question Posted:
Students also viewed these Engineering questions
-
A steady stream of equimolar N2 and CO2 mixture at 100 kPa and 18C is to be separated into N2 and CO2 gases at 100 kPa and 18C. Determine the minimum work required per unit mass of mixture to...
-
Journalize the 10 transactions for your company using excel (must include descriptions). Your opening entry should include the following 4 items: Bank $10,000 Supplies $5,000 Bank Loan $20,000...
-
A total of 2,000 gallons of 70 wt% ethanol in water, having a specific gravity of 0.871, is to be separated at 1 atm in a batch rectifier operating at constant distillate composition with a constant...
-
What is a buy-sell agreement, and how does life insurance facilitate it?
-
The government places a tax on the purchase of socks. a. Illustrate the effect of this tax on equilibrium price and quantity in the socks market. Identify the following areas both before and after...
-
The financial statements of Marks and Spencer plc (M&S) are available at the books companion website or can be accessed at corporate.marksandspencer....
-
Find the LCM of 24 and 90 by listing multiples and choosing the smallest common multiple.
-
Orwell Company accumulates the following adjustment data at December 31. 1. Services provided but not recorded total $1,420. 2. Supplies of $300 have been used. 3. Utility expenses of $225 are...
-
Future value of the following ordinary annuities A) Future value of 800 each month for 4 years at a nominal rate of 8 per cent compounded semiannual. Do not round intermediate calculations round your...
-
A stream that contains a mixture of methane (25% by mol) and carbon monoxide is compressed from 1 bar, 35 to 12 bar. The compressor efficiency is 90%. Treating the mixture as an ideal gas, calculate...
-
Use the data below for the system ethyl propyl ether (1)-chloroform (2) to answer the following questions: a) What is the boiling point of chloroform at 0.5 bar? b) Is this a maximum boiling or...
-
A company is considering buying a new machine for $1,200,000. The new machine will replace the current machine, which can be sold now for $75,000. The company will incur installation costs of...
-
Jose Ruiz manages a construction rm's equipment repair... Jose Ruiz manages a construction firm's equipment repair department. His department is a cost center. Costs for a recent period follow. Jose...
-
The most recent financial statements for GPS, Inc., are shown here: Income Statement Balance Sheet Sales $19,500 Assets $98,000 Debt $52,500 Costs 15,000 Equity 45,500 Taxable income $4,500 Total...
-
Two charged, square plates, separated by a distance of 12.6 cm and each with a side length of 27.1 cm, make up a parallel-plate capacitor. The electric field inside the plates is 1.70x10 3 N/C. If...
-
What volume of a 12.0 M H2SO4 solution would be required to produce 300. mL of 4.0 M H2SO4?
-
1. What are the roles of toroidal and polodial magnetic field on tokamak? 2.Describe the inertial confinement method for nuclear fusion? 3.Why is fusion considered to be safer than fission? 4.How do...
-
What is the difference between a correspondent, respondent, and bankers bank?
-
Kenneth Hubbard has prepared the following list of statements about managerial accounting and financial accounting. 1. Financial accounting focuses on providing information to internal users. 2....
-
Several processes are described. Indicate whether each is reasonably modeled as reversible, and if not, indicate what aspect of the process makes it irreversible. A . The inside of a refrigerator is...
-
A steady-state distillation column is designed to separate benzene from toluene. The separation is nearly enough complete that, for the purposes of designing the reboiler and the condenser, we can...
-
A liquid stream contains 1 lb m /s of the compound at T = 100F and P = 1 atm. It needs to be boiled and heated to P = 1 atm and 175F, as that is the temperature at which it must enter a chemical...
-
Section L: Indirect Pay Plan. You also need to describe the characteristics of your indirect pay plan. Some of the questions to be addressed include the following: Will it be a fixed, flexible, or...
-
[Chapter 6: Advanced drawing techniques] P6-12) Create an orthographic projection of the following object. Draw the three standard views. Place surface roughness symbols on all finished surfaces. 038...
-
Chapter 5, Problem 5/024 Your answer is incorrect. Locate the centroid of the area shown in the figure by direct integration. (Caution: Carefully observe the proper sign of the radical involved.) Set...

Study smarter with the SolutionInn App


