Deduce (5.22) and (5.23) starting from the definition of the electron flux vector and the expression for
Question:
Deduce (5.22) and (5.23) starting from the definition of the electron flux vector and the expression for f1(r, v) given by (5.6), (5.11), (5.12), and (5.13).
Equations
![]()
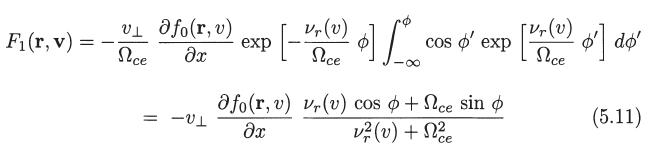
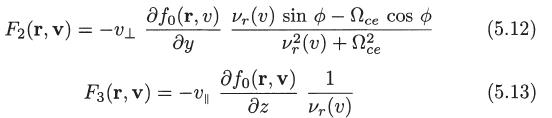
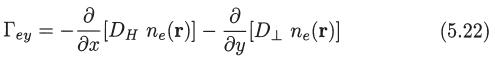
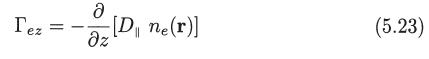
Transcribed Image Text:
fi(r, v) = F₁ (r, v) + F2(r, v) + F3(r, v) (5.6)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
Carol Harris, Ph.D, CPA, is a single taxpayer and she lives at 674 Yankee Street, Durham, NC 27409. Her Social Security number is 793-52-4335. Carol is an Associate Professor of Accounting at a local...
-
A stock has a beta of 1.55 and an expected return of 15 percent. A risk-free asset currently earns 2.2 percent. a. What is the expected return on a portfolio that is equally invested in the two...
-
A firm faces the following average revenue (demand) curve: P = 100 0.01Q Where Q is weekly production and P is price, measured in cents per unit. The firms cost function is given by C = 50Q +...
-
How to obtain a temporary restraining order?
-
Sketch an outline of a pitch for a television series about a forensic accountant?
-
Company produces one type of sunglasses with the following costs and revenues for the year: Total Revenues...............................................................................................
-
Rocky Metalworks, Inc. has a total asset turnover 2.1% and a net profit margin 7.50%. The total debt ratio for the firm is 35.00% a) Calculate Rocky's return on equity. b) Briefly explain how the...
-
Show that (3.30) and (3.31) yield, respectively, (3.37) and (3.38), when r is independent of v for any f 0 (v). Equations. 0=- 4ie 3me T 4ie 3me v3 dfo (v) w + ivr(v) Fce dv dv v3 w + iv, (v) dfo...
-
Analyze the heat flow problem in a weakly ionized plasma immersed in an externally applied magnetostatic field B 0 and derive expressions for the heat flow vector q e and for the components of the...
-
What is an ideal solution? Comment on the volume change, enthalpy change, entropy change, and chemical potential change during the formation of ideal and nonideal solutions.
-
Accountants can report on compliance with laws and regulations in conjunction with an audit engagement or as a separate engagement. What is the difference in the objective of reporting on compliance...
-
A) Use a fixed-point iteration method to determine a solution accurate to within 102 for x-3x-3=0 on [1, 2]. Use po = 1. B) Use the Bisection method for e = 10 find the approximate value of 3.
-
(a) Suppose the power series anz" has radius of convergence R. Show that, for n=0 ol> R, the sequence (an) is unbounded. 8 8 7=0 convergence R=1. 8 (b) If an converges and lan diverges, show that the...
-
The following diagram (The chart is posted in course content) shows the interest each descendant would receive in Howard's estate. All descendants are living except as noted. 25 POINTS TOTAL a. Is...
-
An athlete has 13 % by mass body fat. What is the weight of fat,in pounds, of a 76-kg athlete? Express your answer to twosignificant figures and include the appropriate units.
-
How do regulations regarding barriers to entry and the scope of permitted activities affect the charter value of financial institutions?
-
The test statistic in the NeymanPearson Lemma and the likelihood ratio test statistic K are intimately related. Consider testing H 0 : = 0 versus H a : = a , and let * denote the test statistic...
-
Discuss the following statement: If the temperature of the system increased, heat must have been added to it.
-
Oxygen reacts with solid glycylglycine C 4 H 8 N 2 O 3 to form urea CH 4 N 2 O, carbon dioxide, and water: 3O 2 (g) + C 4 H 8 N 2 O 3 (s) CH 4 N 2 O(s) + 3CO 2 (g) + 2H 2 O(l) At T = 298 K and 1.00...
-
Identify the reagents that you would use to achieve each of the following transformations: a. b. Br Br
-
Samuel plans to deposit $350 at the beginning of every month into a bank account. The bank account has an interest rate of 6% per year. How much will Samuel have in the account at the end of 2 years?...
-
The Assembly Department of ByteSize, Inc., manufacturer of computers, incurred $250,000 in direct material costs and $90,000 in conversion costs. The equivalent units of production for direct...
-
Reference the Level 1 DFD below to answer the following question. Process 2.0: Generate Reports, would include what from the list below? Order 1.0 Order Customer Receipt Orders Cookies Smart Cookie...

Study smarter with the SolutionInn App


