Starting from the expression of the total energy carried by the lattice vibrations in Eq. (6.60), show
Question:
Starting from the expression of the total energy carried by the lattice vibrations in Eq. (6.60), show that the heat capacity Cv = (dE/dT)v can be written as: 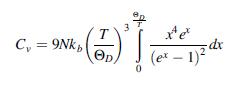
Transcribed Image Text:
T C, = 9Nk, Op (et – 1)2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
The total energy carried by the lattice vibrations can be expressed as E 9NkB T3 qpT2 0DT x3ex ...View the full answer

Answered By

Tobias sifuna
I am an individual who possesses a unique set of skills and qualities that make me well-suited for content and academic writing. I have a strong writing ability, allowing me to communicate ideas and arguments in a clear, concise, and effective manner. My writing is backed by extensive research skills, enabling me to gather information from credible sources to support my arguments. I also have critical thinking skills, which allow me to analyze information, draw informed conclusions, and present my arguments in a logical and convincing manner. Additionally, I have an eye for detail and the ability to carefully proofread my work, ensuring that it is free of errors and that all sources are properly cited. Time management skills are another key strength that allow me to meet deadlines and prioritize tasks effectively. Communication skills, including the ability to collaborate with others, including editors, peer reviewers, and subject matter experts, are also important qualities that I have. I am also adaptable, capable of writing on a variety of topics and adjusting my writing style and tone to meet the needs of different audiences and projects. Lastly, I am driven by a passion for writing, which continually drives me to improve my skills and produce high-quality work.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Show that van der Waals equation can be written as a cubic equation in the compressibility factor involving the reduced pressure and reduced temperature as (27 P 27 P2 512 T, (T+1) z2- - 1) z - Z3 ST...
-
Show that the secular equation (9.82) can be written as
-
Show that the group velocity can be written as dv = v - di Og
-
Pepsin is the principal digestive enzyme of gastric juice. A 1.40 g sample of pepsin is dissolved in enough water to make 4.50 mL of solution. The osmotic pressure of the solution is found to be...
-
Fellowes and Associates Chartered Accountants is a successful mid-tier accounting firm with a large range of clients across Canada. In 2011, Fellowes and Associates gained a new client, Health Care...
-
A loom operator in a textiles factory earns $16 per hour. By contract, the employee earns $20 for overtime hours. The operator worked 45 hours during the first week of May, instead of the usual 40...
-
What conditions are necessary for a risk-free asset to be free of risk? Provide an example. Is it really risk-free?
-
Ten weeks of data on the Commodity Futures Index are: 7.35 7.40 7.55 7.56 7.60 7.52 7. 52 7.70 7.62 7.55 a. Construct a time series plot. What type of pattern exists in the data? b. Use trial and...
-
Answer: The probability that the sample mean number of TV sets is between 2.5 and 3 is 0.0390. Part: 2/5 Part 3 of 5 (c) Find the 40th percentile of the sample mean. Round the answer to at least two...
-
Giselle Schmidt is an analyst for Allied Medical Waste and she needs to prepare a cash budget in order to determine the optimal time to purchase new Medical Waste Transport Vehicles costing $63,000...
-
Plot the shapes of the optical and acoustic branches in the dispersion relation for four different ratios of masses: and 1. Show that, in the case of two identical atoms, there is actually only one...
-
In Sect. 6.1.4, we calculated the ratio of the displacement amplitudes A and B for the long wave limit (k ! 0) for both the optical and acoustic phonon branches and then determined the displacement...
-
Add the attribute onload="SelectImage();" to the BODY element of your slides.html page, so that it randomly selects from the images and displays that image when the page loads. Verify that your page...
-
1.If the local government invests in a project which has a 20 years useful life and initial investment of 2.25 million dollars and the yearly benefit is $250,000, with a maintenance cost (starting in...
-
Clarist Inc. issued 4 0 , 0 0 0 shares of $ 3 par common stock at $ 1 3 . 9 0 per share at the start of the year. In the last month of the year, they bought back 1 0 0 common shares to use as holiday...
-
Pharoah Ltd. began operations on January 2, 2024. During the year, the following transactions affected shareholders' equity: 1. Pharoah's articles of incorporation authorize the issuance of 2.5...
-
Harris Fabrics computes its plantwide predetermined overhead rate annually on the basis of direct labor-hours. At the beginning of the year, it estimated that 44,000 direct labor-hours would be...
-
provide an example of statutory merger& statutory consolidation? and what are some advantages and disadvantages of each statutory merger& statutory consolidation?
-
Search the Web using the key words:"best practices, project management." What did you find? How might this information be useful to a project manager?
-
Havel says the grocer doesnt believe what is on the sign and indeed, he says the grocers customers will barely notice it. But Havel maintains that the sign serves a specific function. How would you...
-
Make a graph of [Ag + ], [AgOH(aq)], [CN - ], and [HCN] as a function of pH in a saturated solution of AgCN. Consider the following equilibria and do not consider activity coefficients. Find the pH...
-
Difference plot. A solution containing 3.96 mmol acetic acid plus 0.484 mmol HCI in 200 mL, of 0.10 M KC1 was titrated with 0.490 5 M NaOH to measure K. for acetic acid. (a) Write expressions for the...
-
Difference plot. A solution containing 3.96 mmol acetic acid plus 0.484 mmol HCI in 200 mL, of 0.10 M KC1 was titrated with 0.490 5 M NaOH to measure K. for acetic acid. (a) Write expressions for the...
-
Your client, Hyun Bin, has an offer to come to Canada to work as a financial auditor to work for Deloitte. The position will be full-time and will last at least one year. He has just graduated from...
-
help with this Requirement 1. Compute the breakeven point in units, assuming that the company achieves its planned sales mix.Begin by determining the sales mix. For every 1 deluxe unit(s)...
-
Withdrawals from an Oregon 529 College Savings Network Account, used to pay for qualified expenses, will not be taxed. Qualified expenses include: Question 49Select one: a. Expenses for tuition,...

Study smarter with the SolutionInn App


