A reversible process in a piston/cylinder is shown in Fig. P6.7. Indicate the storage change u 2
Question:
A reversible process in a piston/cylinder is shown in Fig. P6.7. Indicate the storage change u2 − u1 and transfers 1w2 and 1q2 as positive, zero, or negative.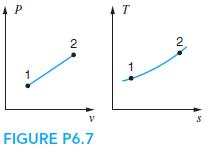
Transcribed Image Text:
2. FIGURE P6.7
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Question Question Description A reversible process in a ...View the full answer

Answered By

Tarique Anwar Khan
I am teaching in an engineering college from last 7 years.
I was in Shivneri polytechnic Junnar from 2013 to 2014. then i have joined to MMANTC, Malegaon as an assistant professor in department of mechanical engineering in 2014 and currently working there.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A reversible process in a steady flow of air with negligible kinetic and potential energy changes is shown in Fig. P7.4. Indicate the change h e h i and transfers w and q as positive, zero, or...
-
A closed gaseous system undergoes a reversible process in which 30 kJ of heat is rejected and the volume changes from 0.14 m 3 to 0.055 m 3 The pressure is constant at 150 kPa. Determine (a) the...
-
A reversible process in a piston/cylinder is shown in Fig. P6.8. Indicate the storage change u 2 u 1 and transfers 1w 2 and 1q 2 as positive, zero, or negative. P 1 FIGURE P6.8 2. 2.
-
O 00:29:33 4. Let an be a convergent series and b, be a soquence such that 0 <2+ a, < b Which of the following statements are true? 10 I) The convergence or divergence of cannot be concluded. Im (2+...
-
You listen to a debate between two politicians discussing the economic health of the United States. One politician says that the average income of all workers in the United States is $72,235; the...
-
What Key Differences in Gendered Communication Does a person Encounter Regularly in their personal, social, familial, academic, and social areas of their Life?
-
One department of Wamego Technology Company manufactures basic handheld calculators. Various materials are added at different stages of the process. The outer front sheet and the carrying case, which...
-
Assume Max convinces Michelle to let him serve as the review partner for all audits and Michelle will serve as the engagement partner on all audits. Do you see any problems with this approach from an...
-
A taxpayer who has a Section 6651(a)(1) penalty has done which of the following? Select one: a. Failed to disclose an uncertain tax position and are due to pay a penalty as a consequence of...
-
Schumann Shoe Manufacturer is considering whether or not to refund a $70 million, 10% coupon, 30-year bond issue that was sold 8 years ago. It is amortizing $4.5 million of flotation costs on the 10%...
-
Liquid water at 20C, 100 kPa is compressed in a piston/cylinder without any heat transfer to a pressure of 200 kPa. How do the properties (T, v, u, and s) change (increase, stay about the same, or...
-
Air at 290 K, 100 kPa in a rigid box is heated to 325 K. How do the properties (P, v, u, and s) change (increase, stay about the same, or decrease)?
-
In problem solve the given differential equation by using the substitution u = y'. x 2 y'' + (y') 2 = 0
-
Windhoek Mines, Limited, of Namibia, is contemplating the purchase of equipment to exploit a mineral deposit on land to which the company has mineral rights. An engineering and cost analysis has been...
-
You have the opportunity to purchase a suburban office property in Chicago. The property is subdivided into individual tenant spaces that total 15860 square feet. The average annual rent per square...
-
Brown pays $90,000 to the Tripper Sprung Partnership in exchange for 30% ownership. Before the admission of Brown, the other partners had the following capital accounts and profit and loss sharing...
-
Which of the tenets in Howard Gardner's (Harvard) recommendations regarding "How Education Changes" describes what students need to learn in a globalized world related to the need for students to be...
-
4. (8 points) Determine if each of the integrals below is an improper integral. If it is not improper, just write not improper. If it is improper, write it as a limit or sum of limits. Do not...
-
John Petosa, a simple Professor of Practice at Syracuse University, has a lifelong dream of owning a winery. He has studied the soils and topography of both New York State and California. It is...
-
At 298.15 K, G f (HCOOH, g) = -351.0 kJ mol -1 and G f (HCOOH, l) 361.4 kJ mol -1 . Calculate the vapor pressure of water at this temperature.
-
In this problem, you will calculate the differences in the chemical potentials of ice and super cooled water, and of steam and superheated water, all at 1 atm pressure shown schematically in Figure...
-
Calculate the vapor pressure of a droplet of benzene of radius 1.25 10 8 m at 38.0C in equilibrium with its vapor. Use the tabulated value of the density and the surface tension at 298 K from...
-
Minden Company is a wholesale distributor of premium European chocolates. The company's balance sheet as of April 30 is given below: MINDEN COMPANY Balance Sheet April 30 Assets Cash Accounts...
-
mr and mrs c shift unearned income of $27,000 in the beginning of 2021 to their son (age 24). their top marginal tax rate is 24% since they have taxable income of $180,000 a year. how much will they...
-
1. What is the purpose of accounting in society? 2. What are the four basic financial statements of a business?

Study smarter with the SolutionInn App


