Use Eq. 12.32 to solve for (T/P)s in terms of T, v, C p , and
Question:
Use Eq. 12.32 to solve for (∂T/∂P)s in terms of T, v, Cp, and αp. How large a temperature change does water at 25◦C (αp = 2.1 × 10−4 K−1) have when compressed from 100 kPa to 1000 kPa in an isentropic process?
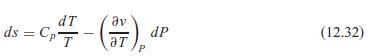
Transcribed Image Text:
dT ds = Cp av dP (12.32) aT
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 30% (13 reviews)
Starting with Eq 1232 ds CpT dT pV dP where s is entropy T is temperature P is pressure v is specifi...View the full answer

Answered By

Anik Kumar Bosu
I am expert in Maths, Physics, Chemistry and Biology. I also teach student from 3 years. Students becomes satisfied by my teaching. So, do not worry.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
2 m' of an ideal gas is compressed from 100 kPa to 200 kPa. As a result of the process, the internal energy of the gas increases by 10 kJ, and 140 kl of heat is lost to the surroundings. What is most...
-
Air is compressed from 100 kPa and 40C. The input power to the adiabatic compressor is 20 kW. Determine the mass flow rate of air through the compressor assuming the air undergoes a quasi-equilibrium...
-
Air is compressed from 100 kPa and 310 K to 1000 kPa in a two stage compressor with intercooling between stages. The intercooler pressure is 350 kPa. The air is cooled back to 310 K in the...
-
Cindy Bagnal, the manager of Cayce Printing Service, has provided you with the following aging schedule for Cayce's accounts receivable: Cindy indicates that the $126,700 of accounts receivable...
-
What is the difference between "dollarization" and a currency board?
-
Match List-I (parts) with dist-II (Method of holding on a lathe) and select the correct answer using the codes given below the lists : List-I (parts) A. Cylindrical parts B. Non-cylindrical parts C....
-
How can one be a leader as a staff nurse?
-
Refer to P2-5. Apple Inc., headquartered in Cupertino, California, designs, manufactures, and markets mobile communication and media devices, personal computers, and portable digital music players...
-
Interest rates in Canada and in major foreign economies A. tend to move in opposite directions. B. are uncorrelated or very weakly correlated. OC. tend to move in the same direction. D. are the same...
-
Dean reads in his physics book that when two people pull on the end of a rope in a tug-of-war, the forces exerted by each on the other are equal and opposite, according to Newton's third law....
-
Use Eq. 12.34 to derive an expression for the derivative (T/v)s. What is the general shape of a constant s process curve in a Tv diagram? For an ideal gas, can you say a little more about the shape?...
-
Use Table B.3 to find the speed of sound for carbon dioxide at 2500 kPa near 100C. Approximate the partial derivative numerically.
-
There is no such thing as a typical organization or management job therefore it is pointless attempting to theorize about them. To what extent would you agree with this view? Justify your answer.
-
Suppose that a fund that tracks the S&P has a mean E(R M ) = 10% and standard deviation M = 11%, and suppose that the T-bill rate R f = 5%. Answer the following questions about efficient portfolios:...
-
Esquire Comic Book Company had income before tax of $1,550,000 in 2024 before considering the following material items: 1. Esquire sold one of its operating divisions, which qualified as a separate...
-
1. What happen on covid 19 nowadays? What is the action of the government about increasing of the cases of covid-19? What is the rules and regulations Implemented under community quarantine? What is...
-
The real risk free rate if interest is 4% (k*), Inflation Premium expected for the next 10 years is 3 % and the Maturity Risk Premium is equal to 0.1(t-1)% where "t" is equal to a security's maturity...
-
Recent coverage of Donald Trump's increasingly outrageous statements in pursuit of the Republican nomination for president has led many to declare that U.S. politics needs reform. Do you agree with...
-
In Section 3.2, we analyzed the heights of the starting five players on each of two men's college basketball teams. The heights, in inches, of the players on Team II are 67, 72, 76, 76, and 84....
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
Draw a reasonable mechanism for the following transformation. NaOH, H,0 Heat
-
Predict the major product for each of the following transformations. (a) (b)
-
Propose an efficient synthesis for each of the following transformations. (a) (b) (c) (d) OEt EtO
-
provide clear, neat, and complete solutions step by step for the given problem. Thanks!
-
What are the potential issues involving funding requirements and revenue streams from three very different businesses - fixed investments in the meat business in South America based in Colombia and...
-
Please assist in finding the correct solution. In a recent 5-year period, mutual fund manager Diana Sauros produced the following percentage rates of return for the Mesozoic Fund. Rates of return on...

Study smarter with the SolutionInn App


