Assume that the packing of spherical atoms in crystalline metals is the same for Li, Na, and
Question:
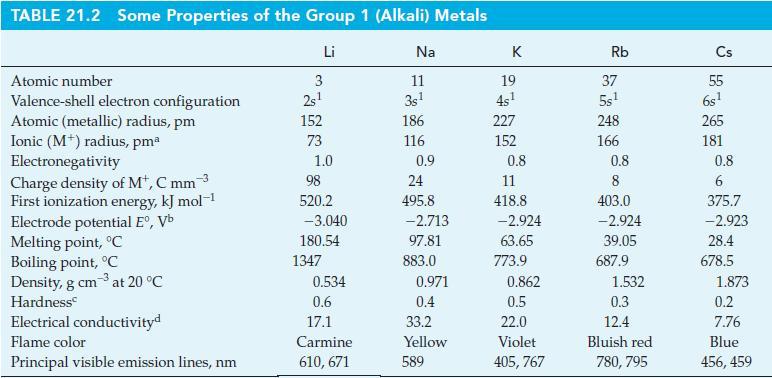
Assume that the packing of spherical atoms in crystalline metals is the same for Li, Na, and K, and explain why Na has a higher density than both Li and K. Use data from Table 21.2.
Table 21.2
Transcribed Image Text:
TABLE 21.2 Some Properties of the Group 1 (Alkali) Metals Li Atomic number Valence-shell electron configuration Atomic (metallic) radius, pm Ionic (M) radius, pmª Electronegativity Charge density of M+, C mm 3 First ionization energy, kJ mol-1 Electrode potential Eº, Vb Melting point, °C Boiling point, °C Density, g cm3 at 20 °C Hardness Electrical conductivityd Flame color Principal visible emission lines, nm 3 2s¹ 152 73 1.0 98 520.2 -3.040 180.54 1347 0.534 0.6 17.1 Carmine 610, 671 Na 11 3s¹ 186 116 0.9 24 495.8 -2.713 97.81 883.0 0.971 0.4 33.2 Yellow 589 K 19 4s¹ 227 152 0.8 11 418.8 -2.924 63.65 773.9 0.862 0.5 22.0 Violet 405, 767 Rb 37 5s¹ 248 166 0.8 8 403.0 -2.924 39.05 687.9 1.532 0.3 12.4 Bluish red 780, 795 Cs 5.5 6s¹ 265 181 0.8 6 375.7 -2.923 28.4 678.5 1.873 0.2 7.76 Blue 456, 459
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Assume that each sequence converges and find its limit. 2,2 + 2 + 2 - 1 2 + 1' 2 2 + 2 + 1 1 2 + 1 - 2
-
In Exercises 125128, determine whether each statement is true or false. If the statement is false, make the necessary change(s) to produce a true statement. In V2 = In 2 2
-
(A) Use data from Table 19.1 to predict the probable products when Pt electrodes are used in the electrolysis of KI(aq). Table 19.1 (B) In the electrolysis of AgNO 3 (aq), what are the expected...
-
If $4000 is deposited into an account paying 3% interest compounded annually and at the same time $2000 is deposited into an account paying 5% interest compounded annually, after how long will the...
-
Using the information from PE 12-50, make the necessary journal entry to record the purchase of this held-to-maturity security.
-
Raul Fletes needed money to buy lawn equipment. He borrowed $500 for seven months and paid $53.96 in interest. What was the rate of interest?
-
Three bases of position power are ____________. (a) reward, expertise, and coercive power (b) legitimate, experience, and judgment power (c) knowledge, experience, and judgment power (d) reward,...
-
Ralph Henwood was paid a salary of $64,600 during 20-- by Odesto Company. In addition, during the year Henwood started his own business as a public accountant and reported a net business income of...
-
ABC Company has the following product costing information for the month of June: Total manufacturing costs $ 150,000 Work in process, June 1 $ 80,000 Work in process, June 30 $ 85,000 Finished goods,...
-
The reaction of borax, calcium fluoride, and concentrated sulfuric acid yields sodium hydrogen sulfate, calcium sulfate, water, and boron trifluoride as products. Write a balanced equation for this...
-
The dissolution of MgCO 3 (s) in NH 4 + (aq) can be represented as Calculate the molar solubility of MgCO 3 in each of the following solutions: (a) 1.00 M NH 4 Cl(aq); (b) A buffer that is 1.00 M NH...
-
Which is not an example of convection current? (a) A moving charged belt (b) Electronic movement in a vacuum tube (c) An electron beam in a television tube (d) Electric current flowing in a copper...
-
Fullarton tachometer a. produces light pulses intermittently b. has high output and is insensitive to temperature c. frequently used in velocity pickups d. has high sensitivity and frequency range e....
-
Each outcome of an experiment, in the case of a random process, is called a a. sample point b. sample space c. sample function
-
Why is it necessary to introduce fictitious mesh points in the finite difference method of solution?
-
For each equation in Problems 7-18, find three ordered pairs that satisfy the equation, and then use this information to graph each line. \(y=x+5 \)
-
Fill in the Blank. The pressure fluctuation at a point on the surface of an aircraft flying in the air is a(n) _____________ process.
-
Michael Norton (of Harvard) and Dan Ariely (of Duke) did a survey reported in Harvard Magazine (Nov-Dec 2011). They asked respondents to estimate how much of the wealth is held by each quintile...
-
The executor of Gina Purcells estate has recorded the following information: Assets discovered at death (at fair value): Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Stock A has an expected return of 7%, a standard deviation of expected returns of 35%, a correlation coefficient with the market of 0.3, and a beta coefficient of !0.5. Stock B has an expected...
-
A stock had a 12% return last year, a year when the overall stock market declined. Does this mean that the stock has a negative beta and thus very little risk if held in a portfolio? Explain.
-
If investors aversion to risk increased, would the risk premium on a high-beta stock increase by more or less than that on a low-beta stock? Explain. Discuss.
-
Explain why executive compensation plans contain various compensation elements (e.g., salary, bonus, stock options, etc.)?
-
. The functional expense allocation is reported in Part IX of form 990. True or False
-
Explain how to calculate the leasing costs for 2020 and 2021

Study smarter with the SolutionInn App


