At 298 K, f G[CO(g)] = -137.2 kJ mol -1 and K = 6.5 x 10
Question:
At 298 K, ΔfG°[CO(g)] = -137.2 kJ mol-1 and K = 6.5 x 1011 for the reaction CO(g) + Cl2(g) ⇌ COCl2(g). Use these data to determine ΔfG°[COCl2(g)], and compare your result with the value in Appendix D.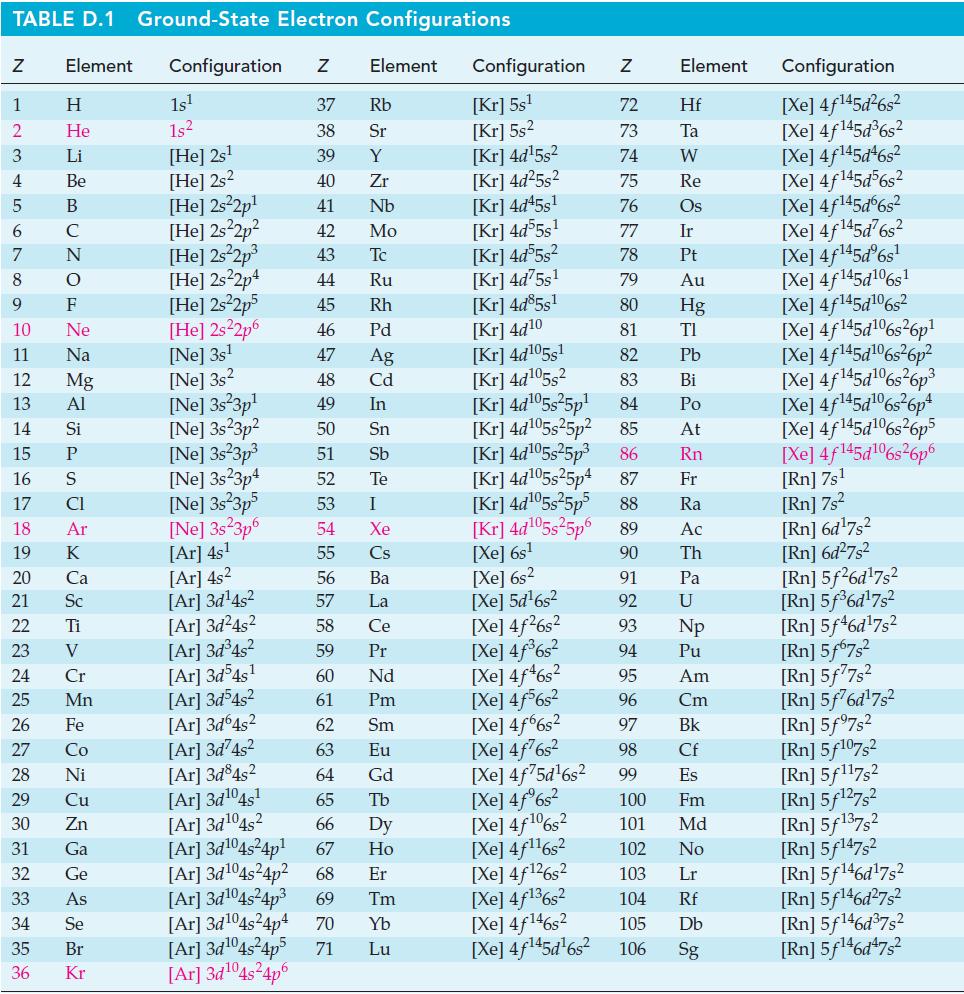
Transcribed Image Text:
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 5 6 7 8 9 HIG&LUZON SJE ZE> 0 ≤ 2 8 2 3 5 3 3 2 2 5 2 He 10 11 12 13 14 15 16 17 18 19 20 21 Sc 22 23 24 25 Mn Mg 26 27 28 Ni 29 30 31 32 33 34 35 36 Zn Ga Ge 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [He] 2s²2p5 [He] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s 3p¹ [Ne] 3s23p² 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb 52 Te 53 54 55 56 57 58 59 60 61 62 [Ar]3d²4s² 63 [Ar] 3d845² 64 [Ar]3d¹04s¹ 65 [Ar] 3d¹04s2 66 67 [Ar]3d¹04s²4p¹ [Ar] 3d¹04s²4p² 68 Er [Ar] 3d¹04s²4p³ 69 Tm 70 Yb Lu [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s²3p5 [Ne] 3s 3p6 [Ar] 4s¹ [Ar] 4s² [Ar] 3d¹4s² [Ar] 3d²4s² [Ar] 3d³4s² [Ar]3d54s¹ [Ar] 3d³4s² [Ar] 3d64s² Element [Ar] 3d¹04s²4p4 [Ar] 3d¹04s²4p5 71 [Ar] 3d¹04s²4p6 I Xe Cs Ba La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Configuration Z [Kr] 5s¹ [Kr] 5s² [kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [kr] 4d55s¹ [kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [Kr] 4d105s1 [kr] 4d¹05s² [Kr] 4d¹05s²5p¹ [kr] 4d¹05s25p² [kr] 4d¹05s²5p³ [kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f²6s² [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [Kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f¹45d²6s² [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d¹7s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
To determine fG for the reaction COg Cl2g COCl2g using the given information you can use the relatio...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Use the data in Appendix 3 to calculate the equilibrium constant for the reaction Agl(s) Ag+(aq) + I2(aq) at 25C. Compare your result with the Ksp value in Table 16.2.
-
In Chapters 2 (Section 2.8) and 3 (Section 3.7) we analyzed the risk and return of the Orange County portfolio, using the building blocks of zero coupon bonds and duration in this exercise, we...
-
A tabulation of more precise thermodynamic data than are presented in Appendix D lists the following values for H 2 O(l) and H 2 O(g) at 298.15 K, at a standard state pressure of 1 bar. (a) Use these...
-
The United Arab Emirates (UAE) is unique in that expatriates constitute more than 80 per cent of the population. As the country continues to grow and accept foreigners at an astounding pace, Emiratis...
-
Fuqua Companys sales budget projects unit sales of part 198Z of 10,000 units in January, 12,000 units in February, and 13,000 units in March. Each unit of part 198Z requires 4 pounds of materials,...
-
What temperature change from 0oC would increase the speed of sound by 1.0%?
-
Jackson Oil Company used 100 Mcf of gas obtained from Lease A and valued at $8.40/ Mcf for gas injection on Lease B. Assume production taxes are 5% and the royalty on Lease A is a 1/6 RI. a. Give the...
-
Procter & Gamble has been the leading soap manufacturer in the United States since 1879, when it introduced Ivory soap. However, late in 1991, its major rival, Lever Bros. (Unilever), overtook it by...
-
Find the value of x to x = 2x 152 143 116 125 139 140
-
At 1000 K, an equilibrium mixture in the reaction CO 2 (g) + H 2 (g) CO(g) + H 2 O(g) contains 0.276 mol H 2 0.276 mol CO 2 , 0.224 mol CO, and 0.224 mol H 2 O. (a) What is K at 1000 K? (b)...
-
Why is r G such an important property of a chemical reaction, even though the reaction is generally carried out under nonstandard conditions?
-
What is investment casting? What are the advantages of investment casting? Explain why this process is often used to cast turbine blades.
-
The current rate for producing 5 amp fuses at Neary Electric Co, is 250 per hour. A new machine has been purchased and installed that, according to the supplier, will increase the production rate. A...
-
What impact do female and/or minority judges have on collegial courts? In other words, do they lead judges to make "different" decisions?
-
(3) A rectangular enclosure is to be built with brick used as the material for three of the retaining walls and fencing used for the one remaining wall. The enclosure needs to have an area of 540...
-
C# please You have decided that people's middle names have not been getting the attention that they truly deserve. To solve this problem, you will create a C# program that will provide a text box...
-
Based on the article 'Microsoft finishes 19.7B acquisition of Nuance' from biometric update: this merger allows Microsoft to "expand its software and services line further into government and...
-
If the potential difference across a capacitor is doubled, what happens to (a) The charge on the capacitor and (b) The energy stored in the capacitor?
-
Data 9.2 on page 540 introduces the dataset Cereal, which includes information on the number of grams of fiber in a serving for 30 different breakfast cereals. The cereals come from three different...
-
FIFO method (continuation of 17-30). Do Problem 17-30 using the FIFO method of process costing. Explain any difference between the cost per equivalent unit in the Assembly Department under the...
-
Transferred-in costs, weighted average method (related to 17-30 to 17-32). Larsen Company, as you know, is a manufacturer of car seats. Each car seat passes through the Assembly Department and...
-
Transferred-in costs, FIFO method (continuation of 17-33). Refer to the information in Problem 17-33. Suppose that Larsen Company uses the FIFO method instead of the weighted-average method in all of...
-
Lee Corporation manufactures and sells party items. The following representative direct labor hours and production costs are provided for a four-month period: Month Direct Labor Hours Production...
-
Campbell Company is a retail company that specializes in selling outdoor camping equipment. The company is considering opening a new store on October 1, year 1. The company president formed a...
-
Edik and Fedora are married for 5 years. Edik bought a bond in his name and the name of Fedora as co-owners. Who has to pay the tax on the bond interest? Edik has to pay the tax on the bond's...

Study smarter with the SolutionInn App


