Complete and balance the following. Write the simplest equation possible. If no reaction occurs, so state. (a)
Question:
Complete and balance the following. Write the simplest equation possible. If no reaction occurs, so state.
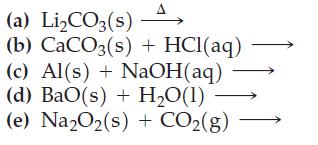
Transcribed Image Text:
(a) Li₂CO3(s) (b) CaCO3(s)+ HCl(aq) (c) Al(s) + NaOH(aq) (d) BaO(s) + H,O(1) (e) Na2O₂(s) + CO₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The balanced equation for the reaction in the image is 2Al...View the full answer

Answered By

Munir Ahmed Jakhro
I am professional Tutor of of Business Courses, I did my four years Bachelor Degree from one of the Top Business schools of World "Institute of Business Administration" in year 2013. Since then I have been working as Tutor of Accounting, Finance tutor on different online platforms like this website. I am have experience of 6 years teaching business courses to students online and offline my professional job at national savings also helped me in accounting understanding .
4.90+
8+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Complete and balance the following equations. Write NR if no reaction occurs. a. I2(aq) + Cl(aq) b. Cl2(aq) + Br(aq) c. Br2(aq) + I(aq) d. Br2(aq) + Cl(aq)
-
Complete and balance the following molecular equations, and then write the net ionic equation for each: (a) HBr(aq) + Ca(OH)2(aq) (b) Cu (OH)2 (s) + HCIO4 (aq) (c) Al(OH)3(s) + HNO3(aq)
-
Q1. How worried are clients and stakeholders in day-to- day product improvement? 2. the industrial corporation Case for Agility "The struggle is not always to the most powerful, nor the race to the...
-
Solve each compound inequality. Graph the solution set, and write it using interval notation. -2x +1> -11 or x+1> 10
-
Foster Enterprises purchased 20% of the outstanding common stock of Novelties, Inc., on January 2, 2012, paying $150,000. During 2012, Novelties reported net income of $20,000 and paid dividends to...
-
Twenty decorative enamel balls cost $12.75 each and are marked up $9.56. a. Find the selling price for each one. b. Find the total amount of margin or markup for the 20 balls.
-
The pattern of authority, influence, and acceptable managerial behavior established at the top of the organization is called ______________. (a) organizational governance (b) agency linkage (c) power...
-
The University of Danville is a private not-for-profit university that starts the current year with $700,000 in net assets: $400,000 unrestricted, $200,000 temporarily restricted, and $100,000...
-
Since managerial accounting does not follow strict reporting requirements like financial accounting, do you see any ethical issues that may arise? Should there be specific reporting requirements? Do...
-
Assuming that water, common reagents (acids, bases, salts), and simple laboratory equipment are available, give a practical method to prepare (a) MgCl 2 from MgCO 3 (s); (b) NaAl(OH) 4 from Na(s) and...
-
A chemist knows that aluminum is more reactive toward oxygen than is iron, but many people believe the opposite. What evidence was provided in this chapter that demonstrates that aluminum is a much...
-
Write pseudocode for the insertion algorithms for linear hashing and for extendible hashing.
-
What is the purpose of experimental modal analysis?
-
Discuss the advantage of using operational deflection shape measurement.
-
What is an electromagnetic shaker?
-
Give two examples of a mechanical vibration exciter.
-
What is phase-shift error? When does it become important?
-
A group of students held their breath as long as possible and recorded the times in seconds. The times went from a low of 25 seconds to a high of 90 seconds, as you can see in the stemplot. Suggest...
-
Suppose the market is semistrong form efficient. Can you expect to earn excess returns if you make trades based on? a. Your brokers information about record earnings for a stock? b. Rumors about a...
-
Heymann Company bonds have 4 years left to maturity. Interest is paid annually, and the bonds have a $1,000 par value and a coupon rate of 9%. a. What is the yield to maturity at a current market...
-
Hooper Printing Inc. has bonds outstanding with 9 years left to maturity. The bonds have an 8% annual coupon rate and were issued 1 year ago at their par value of $1,000. However, due to changes in...
-
Last year Clark Company issued a 10-year, 12% semiannual coupon bond at its par value of $1,000. Currently, the bond can be called in 4 years at a price of $1,060 and it sells for $1,100. a. What are...
-
HW 1 Wishbone Multimedia had the following note payable transactions for 2017 and 2018. June 1, 2017 purchased equipment from Texco costing $18,000 by issuing a one-year, 8% note payable. General...
-
Kingbird Corporation reported net income of $253,200 in 2020 and had 197,000 shares of common stock outstanding throughout the year. Also outstanding all year were 42,000 options to purchase common...
-
Fraser Corporation declares a cash dividend to common shareholders. How does this transaction impact the accounting equation: Assets [Select] Liabilities [Select] and, Stockholders' Equity [Select]

Study smarter with the SolutionInn App


