For the following equilibrium reactions, calculate r G at the indicated temperature. (a) H(g) + I2(g)
Question:
For the following equilibrium reactions, calculate ΔrG° at the indicated temperature.
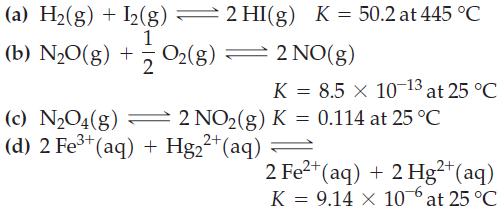
Transcribed Image Text:
(a) H₂(g) + I2(g) 2 HI(g) K = 50.2 at 445 °C 1 (b) N₂0(g) + O2(g) 2 = 2 NO(g) K = 8.5 × 10-13 at 25 °C (c) N₂O4(g) 2 NO₂(g) K = 0.114 at 25 °C (d) 2 Fe³+ (aq) + Hg₂²+ (aq) 2+ 2 Fe²+ (aq) + 2 Hg²+ (aq) K = 9.14 X 10 at 25 °C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
From the Gibbs free energy equation Delta G 2303RTlogK T temperature in kelvin R 8314 jmole K equi...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
At a certain temperature the following reactions have the constants shown: Calculate the equilibrium constant Kc for the following reaction at that temperature: S(s) O2(8O2() 4.2 x 102 25(s) + 302(g)...
-
Calculate the equilibrium constant for the acid-base reactions between the following pairs of reactants. a. HCl + H2O b. CH3COOH + H2O c. CH3NH2 + H2O CH3NH3 + H20
-
Calculate the equilibrium constants of the following reactions at 25C from standard potential data: (a) Sn(s) + CuS04 (aq) ~ Cu(s) + SnS04 (aq) (b) Cu2+(aq) + Cu(s) ~ 2 Cu+{aq)
-
Prepare journal entries to record each of the following sales transactions of TFC Merchandising. TFC uses a perpetual inventory system and the gross method. May 1 9 Sold merchandise for $600, with...
-
Danner Company expects to have a cash balance of $45,000 on January 1, 2014. Relevant monthly budget data for the first 2 months of 2014 are as follows. Collections from customers: January $85,000,...
-
A compact speaker puts out 100 W of sound power. (a) Neglecting losses to the air, at what distance would the sound intensity be at the pain threshold? (b) Neglecting losses to the air, at what...
-
Big John Oil Company purchased 100 barrels of oil from JD Operator. The gross value of the oil was $5,000. The severance tax rate was 4%. Give the entry to record revenue for JD, assuming Big John...
-
Consider an employee who does not receive employer-based health insurance and must divide her $700 per week in after-tax income between health insurance and other goods. Draw this workers opportunity...
-
ou are building a skateboard ramp from a piece of wood that is 3.1 meters long. You want the o make an angle of 25 with the ground. To the nearest tenth of a meter, what is the length of the base?...
-
For the reaction 2 SO 2 (g) + O 2 (g) 2 SO 3 (g), Kc = 2.8 x 10 2 M -1 at 1000 K. (a) What is r G at 1000 K? (b) If 0.40 mol SO 2 0.18 mol O 2 , and 0.72 mol SO 3 are mixed in a 2.50 L flask at...
-
At 1000 K, an equilibrium mixture in the reaction CO 2 (g) + H 2 (g) CO(g) + H 2 O(g) contains 0.276 mol H 2 0.276 mol CO 2 , 0.224 mol CO, and 0.224 mol H 2 O. (a) What is K at 1000 K? (b)...
-
The capital of E. Ltd. is as follows: Additional information: Profit (after tax at 35 per cent), 2,70,000; Depreciation, 60,000; Equity dividend paid, 20 per cent; Market price of equity shares, 40....
-
A company manufactures only custom furniture and uses a job order costing system to accumulate costs. Actual direct materials and direct labor costs are accumulated for each job, but a predetermined...
-
Obtain the following variables from Compustat North America's Fundamentals Annual data via WRDS: GVKEY (firm identifier), FYEAR, DLTT, DLC, CEQ, PRCC_F, CSHO, SICH, and SIC for all available...
-
Croy Incorporated has the following projected sales for the next five months: Month Sales in Units April 3 , 5 3 0 May 3 , 9 5 0 June 4 , 5 5 0 July 4 , 1 6 5 August 3 , 9 5 0 Croy s finished goods...
-
Question 1 You will be creating an invoicing system for the sales of PC parts. The file "Assignment_Q1.xlsm" contains the worksheets "Sales Point", "Items Database", and "Voucher Database". The...
-
JAVA LANGUAGE ONLY!!! I AM USING INTELLIJ IDEA COMMUNITY VERSION MAKE A MATRIX CALCULATOR INSTRUCTIONS ARE WRITTEN BELOW SEE 2ND PHOTO TO SEE WHAT SHOULD THE UML DIAGRAM OF THE CODE SHOULD LOOK LIKE...
-
Give several reasons why a conductor would not be a good choice as a dielectric for a capacitor.
-
Reconsider Prob. 1474. In order to drain the tank faster, a pump is installed near the tank exit as in Fig. P1475. Determine how much pump power input is necessary to establish an average water...
-
Standard-costing method, assigning costs. Refer to the information in Exercise 17-24. Suppose Bio Dec determines standard costs of $6.60 per equivalent unit for direct materials and $10.40 per...
-
Transferred-in costs, weighted-average method. Asaya Clothing, Inc. is a manufacturer of winter clothes, It has a Knitting Department and a Finishing Department This exercise focuses on the Finishing...
-
Transferred-in costs, FIFO method. Refer to the information in Exercise 17-27. Suppose that Asaya uses the FIFO method instead of the weighted-average method in all of its departments. The only...
-
Problem 2: Game Theory. Havard Lorentzen and Cha Min-Kyu race as professional speed skaters. They each decide whether or not to skate Dangerously (D) or Safely (S). They decide simultaneously at the...
-
Whispering Winds and Associates is a medium-sized company located near a large metropolitan area in the Midwest. The company manufactures cabinets of mahogany, oak, and other fine woods for use in...
-
Which of the following is not a component of the master budget? O Capital expenditures budget Selling and administrative expense budget Cash budget Ending inventory and cost of goods sold budget

Study smarter with the SolutionInn App


