Identify and name the functional groups in each of the following. 0 (a) HO-c - || (b)
Question:
Identify and name the functional groups in each of the following.
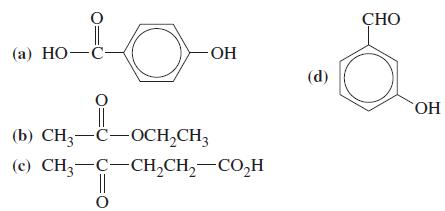
Transcribed Image Text:
0 (a) HO-c -ОН || (b) CH3-C-OCH2CH3 CH3-C-CH2CH2-CO_H (c) || 0 CO,H CHO ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
here are the functional groups in each of the molecules Molecule a Hydroxy group OH This is the OH g...View the full answer

Answered By

Saleem Abbas
Have worked in academic writing for an a years as my part-time job.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Identify the functional groups in each of the following compounds: CH2CH2CH2CH2CH2CH3 Cl OH CH2CH2CH CH H2C CH3CH2CH2CH2 CH2CH2CH2CH3
-
Identify the functional groups in each of the following compounds: (a) H3 C--- CH2--- OH (b) (c) (d) (e) CH3CH2CH2CH2CHO H3C-N CH2CH CH2 CH3C CCH2COOH
-
Identify all of the functional groups in each of the following compounds: (a) (b) (c) (d) (e) (f) (g) Vitamin D3 HO OMe Aspartame O NH2 NH2 Amphetamine Me Cholesterol HO OCH2CH3 Demerol CH A...
-
In Exercises 912, use the given conditions to write an equation for each line in point-slope form and general form Passing through (4, -7) and perpendicular to the line whose equation is x - 2y - 3 =...
-
Saturday Shoppers, Inc., shows the following information in its accounting records at year-end: Sales revenue . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Identify the eight e-policies organisations should implement to protect themselves.
-
A ____________ links key goal-related issues with key collaboration issues to come up with general ways by which the firm will manage its affairs. (a) managerial philosophy (b) cultural symbol (c)...
-
Coakwell Company worked on only two jobs during May. Information on the jobs is given below: At the beginning of the year, annual manufacturing overhead (MOH) was budgeted at $3,780,000 and Coakwell...
-
The beginning balance of Retained Earnings for Briggs Company is $332,000. Net income for the month of July is $22,500, and the company paid dividends of $9,000 in July. What is the ending Retained...
-
By name or formula, give one example of each of the following types of compounds: (a) Aromatic nitro compound; (b) Aliphatic amine; (c) Chlorophenol; (d) Aliphatic diol; (e) Unsaturated aliphatic...
-
The functional groups in each of the following pairs have certain features in common, but what is the essential difference between them? (a) Carbonyl and carboxyl (b) Aldehyde and ketone (c) Acetic...
-
The following information relates to the only product sold by Harper Company. Sales price per unit . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $ 45...
-
Following are selected questions from an internal control questionnaire about a companys fixed asset records, additions, disposals, retirements, and depreciation functions. A yes response would...
-
Cite several significant events in the development of contemporary internal auditing in the U.S.
-
You have been assigned to audit the fixed assets of the Johnson Corp., a manufacturer of janitorial supplies. The company maintains a detailed property ledger for all fixed assets. You prepare an...
-
Identify the general and specific laws and regulations addressed in the Single Audit Act Amendment and OMB Circular A-128, "Audits of States, Local Governments, and Non-profit Organizations."
-
Of the following, which would likely be classified as a Type II subsequent event? a. Collection of receivables in amounts substantially different from amounts recorded at the balance sheet date. b....
-
The table shows the numbers of capital prisoners (prisoners on death row) in 2013 in the western U.S. states. a. Find the median. b. Find the interquartile range (showing Q3 and Q1 in the process)....
-
Discrete sample spaces: suppose there are N cable cars in San Francisco, numbered sequentially from 1 to N. You see a cable car at random; it is numbered 203. You wish to estimate N. (See Goodman,...
-
What does the profitability index (PI) measure? What are the PIs for Franchises S and L?
-
What is the payback period? Find the paybacks for Franchises L and S.
-
What is the rationale for the payback method? According to the payback criterion, which franchise or franchises should be accepted if the firms maximum acceptable payback is 2 years, and if...
-
Lunar Entertainment is a provider of cable, Internet, and on-demand video services. Lunar currently sends monthly bills to its customers via the postal service. Because of a concern for the...
-
ACCT2020-301 Class Project Lease vs. Buy - Capital Budgeting BACKGROUND ONE OF EVERY THREE NEW CARS ON THE ROAD is leased, and consumers are confronted frequently with the decision of whether to buy...
-
The Elements Corporation Soru 1. Elements is an upscale university housing complex providing all the amenities of private townhouse living, and a full service 24-hour cafeteria for busy students....

Study smarter with the SolutionInn App


