The standard electrode potential for the reduction of Eu 3+ (aq) to Eu 2+ (aq) is -0.43
Question:
The standard electrode potential for the reduction of Eu3+(aq) to Eu2+(aq) is -0.43 V. Use the data in Appendix D to determine which of the following is capable of reducing Eu3+(aq) to Eu2+(aq) under standard-state conditions: Al(s), Co(s), H2O2(aq), Ag(s), H2C2O4(aq).
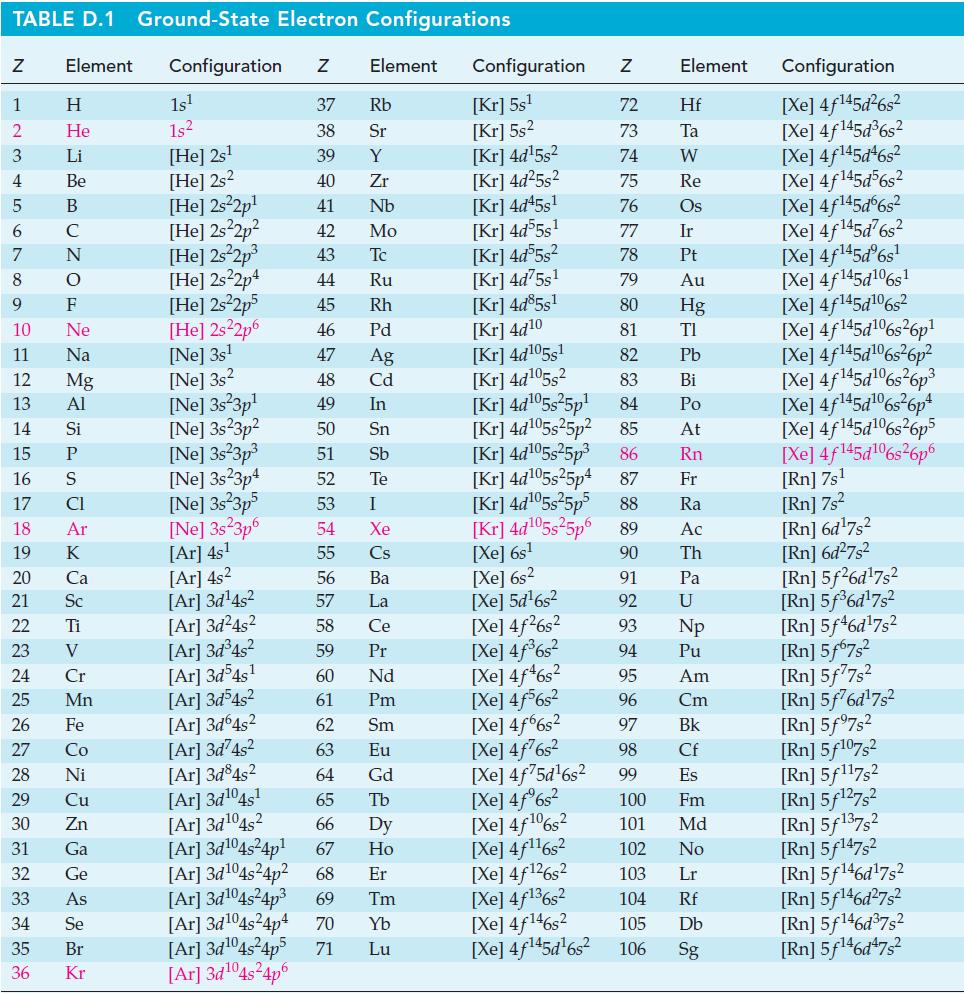
Transcribed Image Text:
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 5 6 7 8 9 HIG&LUZONSUZ SE> 0 ≤ 2 3 2 3 5 3 3 2 2 5 2 He 10 11 12 13 14 15 16 17 18 19 20 21 Sc 22 23 24 25 Mn Mg 26 27 28 Ni 29 30 31 32 33 34 35 36 Zn Ga Ge 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [He] 2s²2p5 [He] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s 3p¹ [Ne] 3s23p² [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s²3p5 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb 52 Te 53 54 55 [Ar] 4s² 56 [Ar]3d¹4s² 57 [Ar]3d²4s² 58 [Ar]3d³4s² 59 [Ar]3d54s¹ 60 61 [Ar]3d³4s² [Ar]3d64s² 62 [Ar]3d²4s² 63 [Ar]3d845² 64 [Ar]3d¹04s¹ 65 [Ar]3d¹04s2 [Ar]3d¹04s²4p¹ [Ar]3d¹04s²4p² [Ar]3d¹04s²4p³ 69 Tm 66 Dy 67 Ho 68 Er [Ar]3d¹04s²4p4 70 Yb Lu [Ar]3d¹04s²4p5 71 [Ar]3d¹04s²4p6 Element [Ne] 3s 3p6 [Ar] 4s¹ I Xe Cs Ba La Ce Pr Nd Pm Sm Eu Gd Tb Configuration Z [Kr] 5s¹ [Kr] 5s² [Kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [Kr] 4d55s¹ [Kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [Kr] 4d105s1 [Kr] 4d¹05s² [Kr] 4d¹05s²5p¹ [Kr] 4d¹05s25p² [Kr] 4d¹05s²5p³ [Kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f²6s² [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [Kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f¹45d²6s² [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d¹7s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Here is a list of the standard electrode potentials for the reduction ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The reaction taking place in an electrochemical cell under standard conditions is Fe 2+ (aq) + Ag + (aq) Fe 3+ (aq) + Ag(s) a. Write two half-equations for this reaction. For each, state whether...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Use the standard electrode potential for A, B, C, D and F given below to answer the questions that follows. The letters do not represent the actual symbols of the elements E Q volts A (aq) 2+ + 2e ...
-
4. Finally, briefly discuss how you could use an empirical econometric model to assess the health and economic growth consequences of a 'fat tax'.
-
How is distribution-channel structure affected by increasing emphasis on the government as a customer and by the existence of state trading agencies?
-
A mixture containing only Al2O3 (FM 101.96) and Fe2O3 (FM 159.69) weighs 2.019 g. When heated under a stream of H2, Al2O3 is unchanged, but Fe2O3 is converted into metallic Fe plus H2O(g). If the...
-
On December 31, Casper, Inc., issued \(\$ 300,000\) of eight percent, ten-year bonds for \(\$ 262,613\), yielding an effective interest rate of ten percent. Semiannual interest is payable on June 30...
-
1. As much as possible, assess the extent to which PetroChinas accounting principles conform to IASB standards. 2. How reliable is your assessment? 3. What further information would help your...
-
Please read Jacobson v. Massachusetts 197 US 11 (1905) and answer these questions 5-7 pages References: https://supreme.justia.com/cases/federal/us/197/11/ What was the question the court was asked...
-
Write the half-cell reactions and the balanced chemical equation for the electrochemical cells diagrammed here. Use data from Table 19.1 and Appendix D to calculate E cell for each reaction. Table...
-
Consider the reaction: Calculate G for this reaction at 25 C under the following conditions: i. P CO2 = 0.112 atm ii. P CCl4 = 0.174 atm iii. P COCl2 = 0.744 atm CO(g) + CCl4(g) = 2 COC1(g)
-
The Auto Clinic is a wholly owned subsidiary of Fast-Check Equipment Company. Fast-Check Equipment sells and leases 4-wheel alignment machines. The usual selling price of each machine is $35,000; it...
-
Consider the following design proposals and financial performance measures for Mighty Manufacturing given in Table 5.39. Both sets of numbers are for 2017 with all possible markets open and plants...
-
Hungry Dragon is a Chinese restaurant specializing in spicy Hunan dishes. It does not have a website, but it has a Twitter account where customers can post comments. On every other week, for a 12...
-
In Peru there are many ruins of the temples and palaces of the Incas, who attained what some historians consider to be the highest pre-European accomplishments in the Americas in agriculture,...
-
Place these markets in the framework that follows. a. Indonesia b. Mozambique c. India d. Bangladesh e. Niger f. Brazil g. Turkey h. Spain i. Singapore j. Nigeria k. Algeria l. Zambia m.Peru n....
-
Using secondary data sources on the Internet, find the following and indicate the appropriate URL (Internet address): a. Number of women who participated in high school basketball last year b....
-
Sport-time produces high-quality basketballs. If the fixed cost per basketball is $3 when the company produces 12,000 basketballs, what is the fixed cost per basketball when it produces 15,000...
-
Briefly describe the following types of group life insurance plans: a. Group term life insurance b. Group accidental death and dismemberment insurance (AD&D) c. Group universal life insurance d....
-
Scully Corporations comparative balance sheets are presented below. Additional information: 1. Net income was $22,630. Dividends declared and paid were $19,500. 2. All other changes in noncurrent...
-
Here are comparative balance sheets for Taguchi Company. Additional information:1. Net income for 2010 was $103,000.2. Cash dividends of $45,000 were declared and paid.3. Bonds payable amounting to...
-
Muldur Corporations comparative balance sheets are presented below. Additional information: 1. Net income was $18,300. Dividends declared and paid were $16,400. 2. Equipment which cost $10,000 and...
-
content area top Part 1 Is the study experimental or observational? A stock analyst selects a stock from a group of twenty for investment by choosing the stock with the greatest earnings per share...
-
A card is being taken out one by one without replacement from a deck of well-shuffled cards till we obtain an ace of spades. The probability of obtaining an ace of spades by the tenth draw is given...
-
Please answers these question with excel : Note. Usea = 0.05 for all tests. The Bank Call Center data shows the data on 70 former employees at one of thebank's call centers. a. Is the average length...

Study smarter with the SolutionInn App


