Use data from Appendix D to determine (a) r H, r S, and r
Question:
Use data from Appendix D to determine
(a) ΔrH°, ΔrS°, and ΔrG° at 298 K and
(b) K at 875 K for the water gas shift reaction, used commercially to produce H2(g): CO(g) + H2O(g) Δ CO2(g) + H2(g).
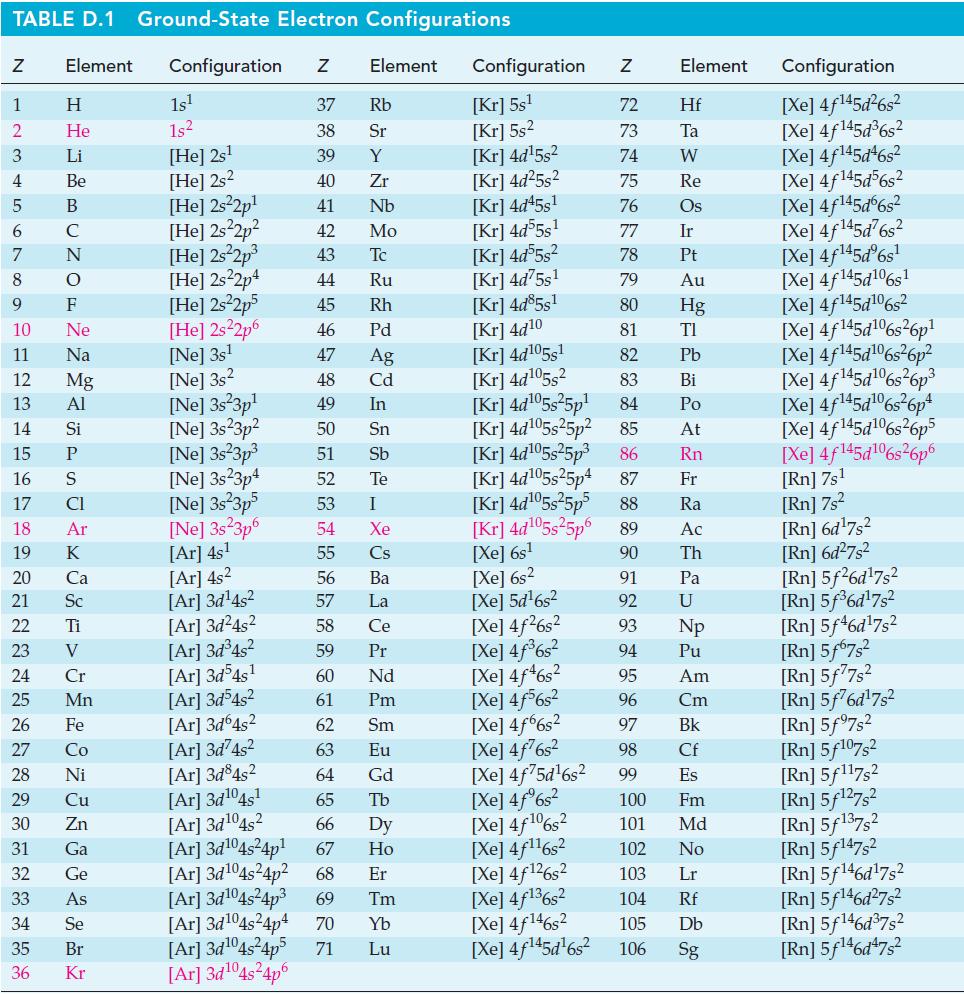
Transcribed Image Text:
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 24 25 27 HIGÅ LUZONS JY SE> 0 ≤ 2 3 2 3 5 3 3 4 8 5 2 Η 29 He 30 Li 31 Be 32 B 33 C F Ne Na 20 Ca Mg 21 Sc Al 22 Ti Si 23 V P CI Ar K 26 Fe 28 Ni Cr Mn Co Cu Zn Ga Ge As 34 Se 35 Br 36 Kr 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [HE] 2s²2p5 [He] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s23p¹ [Ne] 3s23p² [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s 3p5 [Ne] 3s23p6 [Ar] 4s¹ [Ar] 4s² [Ar] 3d¹4s² Element [Ar] 3d¹04s²4p6 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb 52 Te 53 54 55 56 57 58 59 60 61 62 63 64 I Xe Cs Ba La Ce Pr Nd Pm Sm Eu Gd [Ar]3d²4s² [Ar] 3d³4s² [Ar]3d54s¹ [Ar] 3d³4s² [Ar] 3d64s² [Ar] 3d74s² [Ar] 3d845² [Ar] 3d¹04s¹ [Ar] 3d¹04s2 65 Tb 66 67 [Ar] 3d¹04s²4p¹ [Ar] 3d¹04s²4p² 68 Dy Ho Er [Ar] 3d¹04s²4p³ 69 Tm [Ar] 3d¹04s²4p4 70 Yb [Ar] 3d¹04s²4p5 71 Lu Configuration Z [Kr] 5s¹ [Kr] 5s² [kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [kr] 4d55s¹ [Kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [Kr] 4d105s1 [Kr] 4d¹05s² [Kr] 4d¹05s²5p¹ [kr] 4d¹05s25p² [kr] 4d¹05s²5p³ [Kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f²6s² [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f¹45d²6s² [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d¹7s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
a rH rS and rG at 298 K for the water gas shift reaction Using the d...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
In Example 13-3, we dealt with vap H and vap S for water at 100 C. (a) Use data from Appendix D to determine values for these two quantities at 25 C. (b) From your knowledge of the structure of...
-
Use data from Appendix D to establish for the reaction 2 N 2 O 4 (g) + O 2 (g) 2 N 2 O 5 (g): (a) r G at 298 K for the reaction as written; (b) K at 298 K. TABLE D.1 Ground-State Electron...
-
Use data from Appendix D to determine values at 298 K of and K for the following reactions. (The equations are not balanced.) (a) HCl(g) + O(g) (b) FeO3(s) + H(g) (c) Ag(aq) + SO4 (aq) HO(g) + Cl(g)...
-
A particle is thrown vertically upwards into the air. Its height s (in m) above the ground after time t (in seconds) is given by s = 25t 5t 2 (a) What height does the particle reach? (b)What is its...
-
Lager Dental Clinic is a medium-sized dental service specializing in family dental care. The clinic is currently preparing the master budget for the first 2 quarters of 2014. All that remains in this...
-
Suppose that an object can be at any one of n+1 equally spaced points x 0 , x 1 . . . x n . When an object is at location x i , it is equally likely to move to either x i1 or x i+1 and cannot...
-
The Geary Company's income statement and comparative balance sheets as of December 31 of 2019 and 2018 follow: During the year, the following transactions occurred: 1. Sold equipment for \(\$ 9,000\)...
-
Divine Homes manufactures prefabricated chalets in Colorado. The company uses a perpetual inventory system and a job cost system in which each chalet is a job. The following events occurred during...
-
State University Business School (SUBS) offers several degrees, including Bachelor of Business Administration (BBA). The new dean believes in using cost accounting information to make decisions and...
-
Estimate K at 100 C for the reaction 2 SO 2 (g) + O 2 (g) 2 SO 3 (g). Use data from Table 13.8 and Figure 13-10. Table 13.8 Figure 13-10 TABLE 13.8 Equilibrium Constants, K, for the Reaction 2 SO(g)...
-
To establish the law of conservation of mass, Lavoisier carefully studied the decomposition of mercury(II) oxide: (a) Show that the partial pressure of O 2 (g) in equilibrium with HgO(s) and Hg(l) at...
-
Identify whether each of the following items affects the bank or book side of the reconciliation, and indicate whether the amount represents an addition or a subtraction. Bank or Book + or - a....
-
4. Find the positive x-value for the function f (x) = 15 cot (x) where f(x) = 1. x = 3 x = 4 x = 5 x = 6 x = 7
-
What are are examples of ballot box zoning, where Californians have used the ballot box to impact urban growth and planning and place limits on what elected officials and planning agencies can...
-
The Red Fish Company is planning to invest in a major fish hatchery project. There have been two proposals for the investment - Project A and Project B, which are mutually exclusive. The required...
-
You are the hiring manager at a large company in the fashion industry. You are planning to hire a new sales manager and have posted the position in several outlets. You are reviewing applications and
-
(1.) The Taylor Radio Company manufactures and sells ultrasonic radios at P650 each. The variable manufacturing costs per unit are: direct materials = P200, direct labor = P150, FOH = P50, and...
-
A grocery supplier believes that the mean number of broken eggs per dozen is 0.6, with a standard deviation of 0.5. You buy 3 dozen eggs without checking them. a) How many broken eggs do you expect...
-
Prove the result that the R 2 associated with a restricted least squares estimator is never larger than that associated with the unrestricted least squares estimator. Conclude that imposing...
-
Absorption costing and production-volume variance-alternative capacity bases. Earth Light First (ELF), a producer of energy-efficient light bulbs, expects that demand will increase markedly over the...
-
Operating income effects of denominator-level choice and disposal of production-volume variance 1. If ELF sells all 220,000 bulbs produced, what would be the effect on operating income of using each...
-
Cost allocation downward demand spiral Deli One operates a chain of 10 hospitals in the Los Angeles area. Its central food-catering facility, Deliman, prepares and delivers meals to the hospitals. It...
-
The goal of this assignment is to demonstrate that you are able to build a logistics network. You will imagine that you run a company that markets a product on Amazon and you need to stock your...
-
content area top Part 1 A manager estimates the annual after-tax international investment free cash flow from an investment, determines its present value, and then determines it will take to recoup...
-
You are choosing between two projects. The cash flows for the projects are given in the following table ($ million): Project A B Year 0 -$51 - $100 a. What are the IRRS of the two projects? Year 1...

Study smarter with the SolutionInn App


