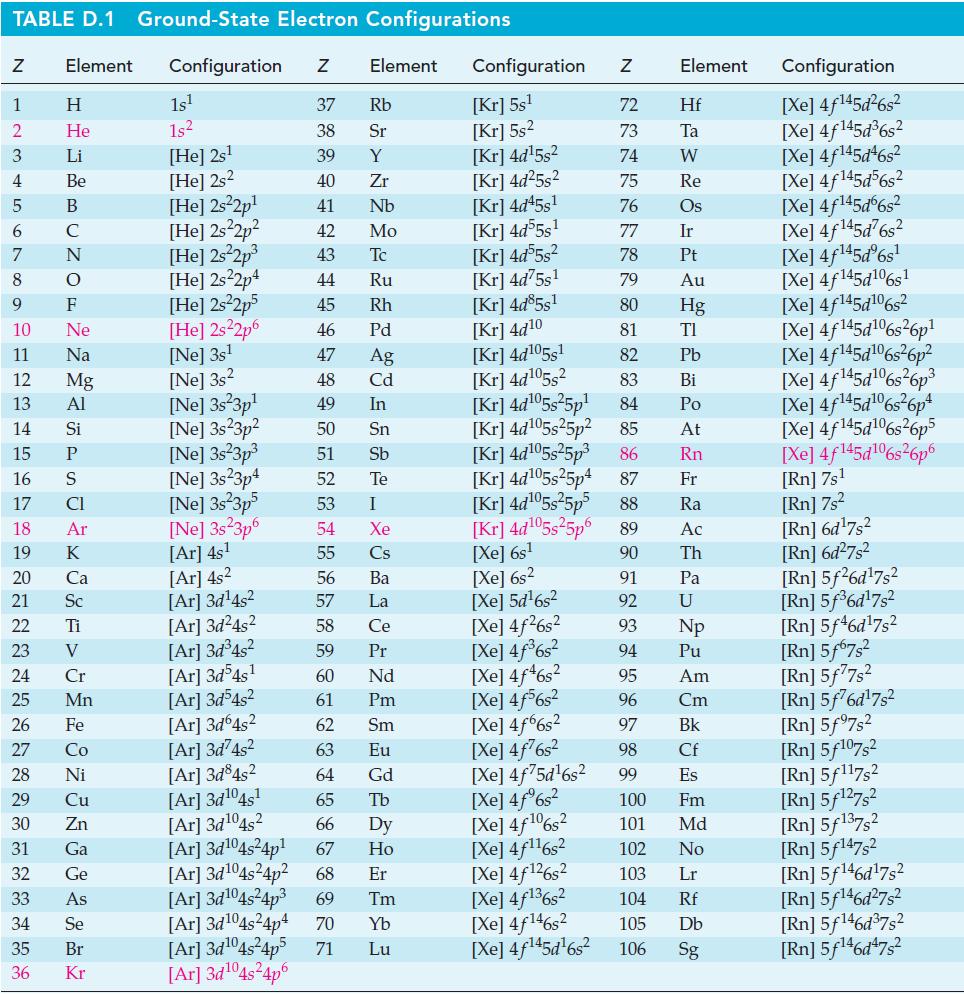
Use data from Appendix D to estimate (a) The normal boiling point of mercury and (b) The
Question:
Use data from Appendix D to estimate
(a) The normal boiling point of mercury and
(b) The vapor pressure of mercury at 25 °C.
Transcribed Image Text:
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 25 27 29 30 HIGÅ LUZONSUZ SE> 0 ≤ 2 3 2 3 5 3 3 2 2 5 2 31 Η 32 He 33 Li Be B C F Ne Na Mg Al 21 Sc 22 Ti 23 V 24 Si P CI Ar 26 Fe K Ca 28 Ni Cr Mn Co Cu Zn Ga Ge As 34 Se 35 Br 36 Kr 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [HE] 2s²2p5 [HE] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s 3p¹ [Ne] 3s23p² [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s 3p5 [Ne] 3523p6 [Ar] 4s¹ [Ar] 4s² [Ar] 3d¹4s² Element [Ar]3d¹04s²4p6 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb Te 52 53 54 55 56 57 58 59 60 61 62 63 64 I Xe Cs Ba La Ce Pr Nd Pm Sm Eu Gd [Ar]3d²4s² [Ar] 3d³4s² [Ar] 3d54s¹ [Ar]3d³4s² [Ar] 3d64s² [Ar] 3d74s² [Ar] 3d845² [Ar] 3d¹04s¹ [Ar] 3d¹04s2 65 Tb 66 67 [Ar] 3d¹04s²4p¹ [Ar] 3d¹04s²4p² 68 Dy Ho Er [Ar] 3d¹04s²4p³ 69 Tm [Ar] 3d¹04s²4p4 70 Yb [Ar] 3d¹04s²4p5 71 Lu Configuration Z [Kr] 5s¹ [Kr] 5s² [kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [kr] 4d55s¹ [kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [Kr] 4d105s1 [kr] 4d¹05s² [kr] 4d¹05s²5p¹ [Kr] 4d¹05s25p² [kr] 4d¹05s²5p³ [kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f²6s² [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f¹45d²6s² [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d47s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a The normal boiling point of a liquid is the temperature at which its vapor pressure equals 1 atmWe ...View the full answer

Answered By

Diksha Bhasin
I have been taking online teaching classes from past 5 years, i.e.2013-2019 for students from classes 1st-10th. I also take online and home tuitions for classes 11th and 12th for subjects – Business Studies and Economics from past 3 years, i.e. from 2016-2019. I am eligible for tutoring Commerce graduates and post graduates. I am a responsible for staying in contact with my students and maintaining a high passing rate.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Find the measure of the indicated angle in the parallelogram. Find mL B. A 51 B D C
-
DE is a midsegment of ABC. Find the value of x. A 6 B. D E X C
-
A steel cylinder with rigid walls is evacuated to a high degree of vacuum; you then put a small amount of helium into the cylinder. The cylinder has a pressure gauge that measures the pressure of the...
-
Using Figure 11.25 and the related text, describe the features of terracing, and list the major alterations to soil and water brought about by this agricultural method. Figure 11.25
-
The budget committee of Widner Company collects the following data for its Westwood Store in preparing budgeted income statements for July and August 2014. 1. Expected sales: July $400,000, August...
-
A 0.50-kg mass oscillates in simple harmonic motion on a spring with a spring constant of 200 N/m. What are (a) The period and (b) The frequency of the oscillation?
-
Bruno Field is jointly owned by Ramsey Company (70% WI), which acts as field operator, and Garza Company (30% WI). There is a 1/6 royalty. The 1/6 royalty is shared proportionally by Ramsey and...
-
Big Blue Rental Corp. provides rental agent services to apartment building owners. Big Blue Rental Corp.'s preliminary income statement for August 2010, and its August 31, 2010, preliminary balance...
-
Given that P(B) = 0.4 and P(A|B) = 0.25, what must the P(A and B) = ? 0.100 0.160 0.416 1.000
-
In an adiabatic process, there is no exchange of heat between the system and its surroundings. Of the quantities S, S surr , and S univ , which one(s) must always be equal to zero for a spontaneous...
-
The synthesis of glutamine from glutamic acid is given by Glu - + NH 4 + Gln + H 2 O. The standard Gibbs energy of reaction* at pH = 7 and T = 310 K is r G' = 14.8 kJ mol -1 . Will this reaction be...
-
You buy a stock option with an exercise price of $50. The cost of the option is $4. If the stock ends up at $56, indicate whether you have a profit or loss with a call option? With a put option?
-
Question 19 ArrayList - Coding Goal: Given the following ArrayList class, which is an ArrayList with a backingArray and a size variable (the same as your homework), implement the addAll(T[] arr, int...
-
You are given the following data for J Corp for January 2020: Purchases of raw materials on account of 70,000; RM inventory, beginning is 13,000; RM, ending inventory balance is 10,000; Indirect...
-
NUBD Co. is planning to market 300,000 units of Product X. The fixed costs are P600,000 and the variable costs are 60% of the selling price. Compute the selling price per unit if the company expects...
-
A product is sold at $100 and its contribution margin percentage is 20%. It also has a fixed cost of $800,000. What is the breakeven sales revenue?
-
Selected transactions of the City of Miser Station General Fund for the 20X1 fiscal year are presented on the following page. All amounts are in thousands of dollars. Prepare the journal entries for...
-
In a computer monitor, electrons are accelerated from rest through a potential difference in an electron gun arrangement (Fig. 16.26). (a) Should the left side of the gun be at (1) a higher, (2) an...
-
Complete problem P10-21 using ASPE. Data from P10-21 Original cost ................................................................. $7,000,000 Accumulated depreciation...
-
Comprehensive variance analysis Kitchen Whiz manufactures premium food processors. The following is some manufacturing overhead data for Kitchen Whiz for the year ended December 31, 2010: Budgeted...
-
Journal entries 1. Prepare journal entries for variable and fixed manufacturing overhead (you will need to calculate the various variances to accomplish this). 2. Overhead variances are written off...
-
Graphs and overhead variances. Fresh, Inc. is a manufacturer of vacuums and uses standard costing. Manufacturing overhead (both variable and fixed) is allocated to products on the basis of budgeted...
-
For Questions 5 and 6 refer to the following information Subgrade Width Station 0+000 centerline top of subgrade elevation Highway vertical grade from 12.0m 867.000m -1.50% Station 0+000 Horizontal...
-
Given the following data for a company: Sales Revenue $36,000 Cost of goods sold 24,080 Gross profit 11,920 Operating expenses 7,120 Net income $4,800 How would cost of goods sold appear on a common...
-
Low Carbohydrate Low Fat Mediterranean Mean -2.9269 -3.1423 -5.4654 Median -2.9500 -3.4000 -5.8500 Standard Deviation 2.02436 2.49835 2.36439 Variance 4.098 6.242 5.590 Minimum -8.50 -7.90 -9.20...

Study smarter with the SolutionInn App


