What is the relationship, if any, between the molecules in each of the following pairs? The relationship
Question:
What is the relationship, if any, between the molecules in each of the following pairs? The relationship may be any of identical structures, constitutional isomers, stereoisomers, or no relationship.
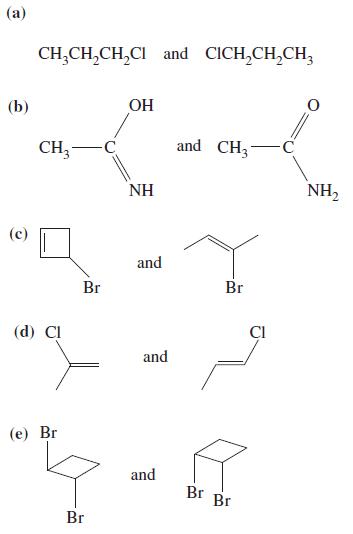
Transcribed Image Text:
(a) (b) (c) CH₂CH₂CH₂Cl and CICH₂CH₂CH3 CH₂ -C (d) Cl (e) Br Br Br OH NH and and and and CH3 Br Br Br CI C O NH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Starting from the top left and going clockwise a CHCHCHCl ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
What is the relationship, if any, between the molecules in each of the following pairs? The relationship may be any of identical structures, constitutional isomers, stereoisomers, or no relationship....
-
Ephedrine has been known in medicine since the Chinese isolated it from natural sources in about 2800 BC. Itsstructure has been known since 1885. Ephedrine can be used as a bronchodilator (a compound...
-
1. The current interest rates are at 8.50%. The bond youre holding pays you $45 semi-annually for the next ten years. What is the present value of your bond, which has a maturity value of $1,000? 2....
-
Write a formula for a function g whose graph is similar to f(x) but satisfies the given conditions. Do not simplify the formula. f(x) = 3x 3x + 2 (a) Shifted right 2000 units and upward 70 units (b)...
-
Assuming the following data, compute: 1. Cash collected from customers 2. Cash paid for wages and salaries 3. Cash paid for inventory purchases 4. Cash paid fortaxes Balance Sheet Income Statement...
-
The September bank statement for Dixon Fence Company shows a balance of $275.25 and a service charge of $7.50. The account register shows deposits of $120.43 and $625.56 that do not appear on the...
-
Explain three ways in which shared leadership can be used in a self-directed work team.
-
A 5.00-F capacitor is initially chmged to a potential of 16.0 V. It is then connected in series with a 3.75-mH inductor. (a) What is the total energy stored in this circuit? (b) What is the maximum...
-
PART A: If Jacob Inc. had a beginning balance of long term notes payable of $645,000, issued new notes payable during the period of $82,000 and had an ending balance of long term notes payable of...
-
How many chiral centers are there in 2,3-dibromopentane? How many different stereoisomers are there?
-
Which chair conformation of cis-1-fluoro-4-methylcyclohexane is lower in energy?
-
The following formulas give the populations of four different towns, A, B, C, and D, with t in years from now. (a) Which town is growing fastest (that is, has the largest percentage growth rate)? (b)...
-
Accurate recording within general ledger control accounts does not necessarily mean that billing, collection, and adjustment transactions will be posted accurately to individual customer accounts....
-
Windek is nearing completion of an audit of the financial statements of Jubilee, Inc. for the year ended December 31, 1999. Windek is currently concerned that subsequent events may require adjustment...
-
An entity has implemented a control procedure which requires that authorized personnel reconcile the total of individual customer accounts receivable with control totals. This control relates to...
-
Statement on Auditing Standards No. 19, "Client Representations," requires that an independent auditor obtain a written representation letter from management, and precludes the auditor from issuing...
-
Consider the graph of \(y=x^{2}\) bounded by the \(x\)-axis and the line \(x=2\). Approximate the area under the curve by using rectangles and right endpoints as described in Problems 47-52. Use...
-
The graphs show the circumferences of heads for a group of men and a group of women. a. If you were describing the men's heads in terms of shape, center, and spread, without comparing them to the...
-
What is the expected payoff of an investment that yields $5,000 with a probability of 0.15 and $500 with a probability of 0.85? Select one: O a. $325 O b. $5,500 O c. $2,750 O d. $1,175
-
What is the primary weakness of sensitivity analysis? What is its primary usefulness?
-
What is the worst-case NPV? The best-case NPV? Use the worst-, base-, and best-case NPVs and probabilities of occurrence to find the projects expected NPV, standard deviation, and coefficient of...
-
Are there problems with scenario analysis? Define simulation analysis, and discuss its principal advantages and disadvantages.
-
Joseph Schumpeter sald that progress occurs through the process of saving and Investment. O True O False
-
The following data are available for the most recent year of operations for Slacker & Sons. The revenue portion of the sales activity variance is $125,000 F. Master budget based on actual sales of...
-
Sanjeev enters into a contract offering variable consideration. The contract pays him $3,000/month for six months of continuous consulting services. In addition, there is a 60% chance the contract...

Study smarter with the SolutionInn App


