Write chemical equations to represent the most probable outcome in each of the following. If no reaction
Question:
Write chemical equations to represent the most probable outcome in each of the following. If no reaction is likely to occur, so state.
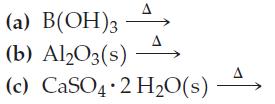
Transcribed Image Text:
(a) B(OH)3 (b) Al2O3(s) (c) CaSO4 2 H₂O(s) A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The chemical equations to represent the most probable outcome in each of the following reaction...View the full answer

Answered By

Divya C
I have completed my graduation in Electronics and Communication Engineering in 2017 but after earning my degree, I began to explore myself, my interests and career I should take up in future. I then realised that my true interest lies in tutoring and mentoring fellow students and not in joining large Tech companies as Engineers. After realizing this I quickly joined a coaching center and started working as tutor in India taking coaching classes for the prestigious IITJEE entrance exam in India. Its been 1.5 yrs in this tutoring career and I simply love it!
0.00
0 Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Write balanced chemical equations to represent the following observations. (In some instances the complex involved has been discussed previously in the text.) (a) Solid silver chloride dissolves in...
-
Use information from the chapter to write chemical equations to represent each of the following: (a) Reaction of rubidium metal with water (b) Thermal decomposition of aqueous KHCO 3 (c) Combustion...
-
Use information from the chapter to write chemical equations to represent each of the following: (a) Reaction of cesium metal with chlorine gas; (b) Formation of sodium peroxide (Na 2 O 2 ); (c)...
-
In Problems 1158, perform the indicated operation, and write each expression in the standard form a + bi. 2 + 3i 1-i
-
Eysser Corporation purchased $50,000 of Hillside Construction Companys 10% bonds at l03 plus accrued interest on February 1, 2011. The bonds mature on April 1, 2018, and interest is payable on April...
-
A Yamaha 88 Portable Grand Keyboard has a cost of $345.58 and is marked up 63% based on cost. Find the selling price and cost of the keyboard.
-
Identify and explain at least four strategies of managerial influence. Give examples of how each strategy may or may not work when exercising influence (a) downward and (b) upward in organizations.
-
In recent years the fast-food chain Wendys International has purchased many treasury shares. From December 28, 2003, to December 31, 2006, the number of shares outstanding has fallen from 115 million...
-
EXAMPLE 1 A property was purchased on 1 January 20X0 for $2m (estimated depreciable amount $1m useful economic life 50 years). Annual depreciation of $20,000 was charged from 20X0 to 20X4 inclusive...
-
A chemical dictionary gives the following descriptions of the production of some compounds. Write plausible chemical equations based on these descriptions. (a) Lead(II) carbonate: adding a solution...
-
Write an equation to represent the reaction of gypsum, CaSO 4 2 H 2 O, with ammonium carbonate to produce ammonium sulfate (a fertilizer), calcium carbonate, and water.
-
Nathan's Athletic Apparel has 1,000 shares of 7%, $100 par value preferred stock the company issued at the beginning of 2011. All remaining shares are common stock. The company was not able to pay...
-
What is the method of isoclines?
-
Define these terms: phase plane, trajectory, singular point, phase velocity.
-
True or False. The Mathieu equation is an autonomous equation.
-
True or False. Nonlinear analysis of a system can reveal several unexpected phenomena.
-
True or False. A singular point corresponds to a state of equilibrium of the system.
-
The graph shows the results of a study done by Guerin et al. (2013) and reported in the New England Journal of Medicine. Patients arriving at emergency rooms who were having trouble breathing were...
-
Prove the following D,(cos x) = - sin x (Hint: Apply the identity cos(A + B) = cos A cos B sin A sin B)
-
Why are convertibles and bonds with warrants typically offered with lower coupons than similarly rated straight bonds?
-
Explain whether the following statement is true or false: Only weak companies issue debentures.
-
Would the yield spread on a corporate bond over a Treasury bond with the same maturity tend to become wider or narrower if the economy appeared to be heading toward a recession? Would the change in...
-
Net Present Value A project has estimated annual net cash flows of $10,000 for ten years and is estimated to cost $42,500. Assume a minimum acceptable rate of return of 20%. Use the Present Value of...
-
Shrey ASSIGNMENT 2) Each of the program budgets must be developed using the information below and using object classification numbers provided 1) This is a time-consuming assignment In your note...
-
Which of the following provisions of the CARES Act does California law conform to? a. Taxpayers were temporarily exempt from the early withdrawal penalty from 401(k) plans. b. There is a five-year...

Study smarter with the SolutionInn App


