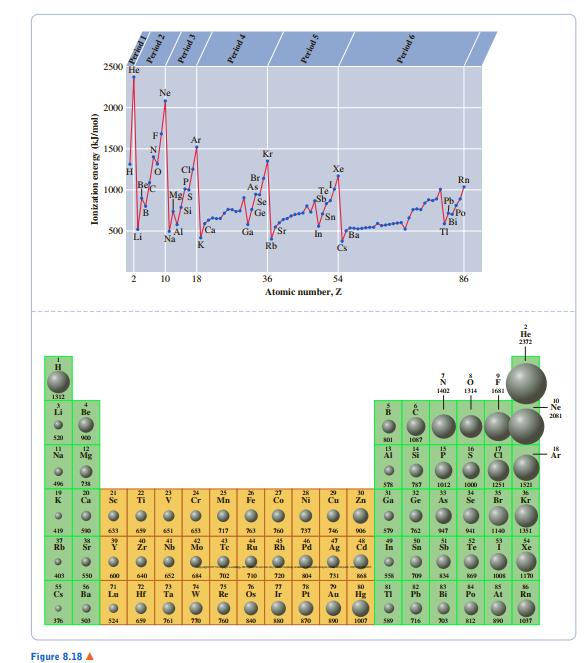
From Figure 8.18, predict the first ionization energy of ununseptium (Z = 117). 2500 He 2000 *
Question:
From Figure 8.18, predict the first ionization energy of ununseptium (Z = 117).
Transcribed Image Text:
2500 He 2000 * 1500 Kr Br As Rn E 1000 Bi S00 Li Ca Sr Na In Ba K Rb Cs 10 18 36 54 86 Atomic number, Z -Ne 吕 550 1008 At 524 730 J0 s40 S0 203 K12 Figure 8.18 A pona Period 2 Period 3 Ionizat ion energy (kJ/mol) Period 4 Perlod 5 つ=2の o 6 Perlod 6 AZ 中 一
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
The ionization energy of ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
From Figure 8.18, predict the first ionization energy of francium (Z = 87). 2500 He 2000 * 1500 Kr Br As Rn E 1000 Bi S00 Li Ca Sr Na In Ba K Rb Cs 10 18 36 54 86 Atomic number, Z -Ne 550 1008 At...
-
The first ionization energy of the chlorine atom is 1251 kJ/mol. Without looking at Figure 8.18, state which of the following values would be the more likely ionization energy for the iodine atom....
-
The first ionization energy of the oxygen molecule is the energy required for the following process: O2 (g) O2+ (g) + e- The energy needed for this process is 1175kJ/mol, very similar to the first...
-
What is a reporting entity?
-
For each of the following, state whether the events created are mutually exclusive and whether they are collectively exhaustive. a. Undergraduate business students were asked whether they were...
-
What rewards are easiest for managers to control? What rewards are more difficult to control?
-
Should forensic accountants engage in covert surveillance?
-
Forten Company, a merchandiser, recently completed its calendar- year 2013 operations. For the year, (1) all sales are credit sales, (2) all credits to Accounts Receivable reflect cash receipts from...
-
Your Mother has been working in a small bookstore for many years. Her sales in the first year were $28,302 and her sales in the last year were $66,425. If the sales grew at an average of 4.43 percent...
-
Create an abstract Student class for Parker University. The class contains fields for student ID number, last name, and annual tuition. Include a constructor that requires parameters for the ID...
-
What property of an atom does nuclear magnetic resonance depend on? What frequency range of the electromagnetic spectrum does NMR use?
-
For eka-lead, predict the electron configuration, whether the element is a metal or nonmetal, and the formula of an oxide.
-
The trials in a binomial distribution are independent. In Exercises 810, determine whether the statement is true or false. If the statement is false, rewrite it as a true statement.
-
Give some applications, where the temptation pattern is used.
-
What do you think are the implementation issues for the AnyUnit BO, when used in the Temptation stable analysis pattern?
-
What role do you think considerations of a child's cultural/racial heritage should play in a custody dispute? What are the potential benefits of giving weight to this factor? What are the potential...
-
Why did the federal government decide to enter the child support field?
-
Assess two different quantitative measures on the temptation traditional model and temptation stable analysis patterns, and explain the differences between each of the measures.
-
Write each expression in Problems 1-3 as the sum or difference of two logarithmic functions containing no exponents. 1. log (x / x + 1) 2. ln [(x + 1)(4x + 5)] 3. log7 (x 3x + 4)
-
Sue Deliveau opened a software consulting firm that immediately paid $2,000 for a computer. Was this event a transaction for the business?
-
Define the terms strong electrolyte and weak electrolyte. Give an example of each.
-
Part 1 a. Both NaCl and MgCl2 are soluble ionic compounds. Write the balanced chemical equations for these two substances dissolving in water.
-
You need to perform gravimetric analysis of a water sample in order to determine the amount of Ag+ present.
-
Post merger earnings per share are affected by all of the following factors, except for A. Acquiring firm's outstanding shares B. Price offered for the target company Number of target firm's...
-
4. Explain the differences in the sampling distributions of x for large and small samples under the following assumptions. Complete parts a and b. a. The variable of interest, x, is normally...
-
The Administrative Procedure Act: O does not apply to federal agencies. O applies to executive branch agencies but not to independent agencies. O applies to independent agencies but not to executive...

Study smarter with the SolutionInn App


