One of the following compounds has a carbonnitrogen bond length of 116 pm; the other has a
Question:
One of the following compounds has a carbon–nitrogen bond length of 116 pm; the other has a carbon–nitrogen bond length of 147 pm. Match a bond length with each compound.
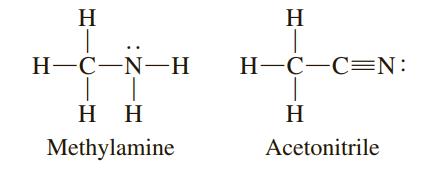
Transcribed Image Text:
H H Н-С—N—Н H-C-C=N: H H H Methylamine Acetonitrile
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
Methylamine 147 pm S...View the full answer

Answered By

Arshad Ahmad
Well, I am really new to tutoring but I truly believe a good student can be a better teacher. I have always been a topper at school. I passed my Chartered Accountancy at a very young age of 23, a rare feat for most of the students. I am really dedicated to whatever work I do and I am very strict regarding deadlines. i am always committed and dedicated to whatever work allotted to me and I make sure it is completed well within deadline and also I try to give my best in whatever I do. Hope we will have a good time studying together.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each of the following compounds has a nitrogennitrogen bond: N2, N2H4, N2F2. Match each compound with one of the following bond lengths: 110 pm, 122 pm, 145 pm. Describe the geometry about one of the...
-
Each of the following compounds has three possible names listed for it. For each compound, what is the correct name and why arent the other names used? a. N2O: nitrogen oxide, nitrogen (I) oxide,...
-
The base peak appears at m/z 105 for one of the following compounds and at m/z 119 for the other two. Match the compounds with the appropriate m/z values for their base peaks.
-
In Exercises 1126, determine whether each equation defines y as a function of x. 4x = y 2
-
You are the manager of a stock portfolio worth $10,500,000. It has a beta of 1.15. During the next three months, you expect a correction in the market that will take the market down about 5 percent;...
-
Calculating OCF Prather, Inc., has sales of $14,200, costs of $5,600, depreciation expense of $1,200, and interest expense of $680. If the tax rate is 35 percent, what is the operating cash flow, or...
-
An investment center is responsible for: a. Investing in long term assets. b. Controlling costs. c. Generating revenues. d. All of the above.
-
The following sample information was obtained by taking four doughnuts per hour for twelve hours from Fawcett Bakerys doughnut process and weighing them: For the data shown above a. Find the x and R...
-
Imagine that a friend tells you that you should not rush to pay off mortgage early because you will lose out on the interest tax deductions you are getting. Discuss the role of amortization of...
-
According to a study several years ago by the Personal Communications Industry Association, the average cell phone user earns $62,600 per year. Suppose a researcher believes that the average annual...
-
Which of the following two compounds has the shorter carbonoxygen bond? H :0: - - H Methanol Formaldehyde
-
Calculate the CCl and CC bond lengths in ethyl chloride, C 2 H 5 Cl, using values for the covalent radii from Table 9.4. How do these values compare with the experimental values: CCl, 177; CC, 155...
-
What information is lost by the use of the information-reducing grammar?
-
A quality control department is testing 25 smartphones from a shipment of 300 smartphones. Describe how this test could be done. Then use technology to generate a list of 25 random numbers from 1 to...
-
A forecasting technique that takes the previous forecast and adds some percentage of the previous forecast's error is called what?
-
The use of which alpha would result in a smoother forecast, a = 0.2 or a = 0.5?
-
In time series analysis, any pattern that regularly repeats itself and is constant in length is referred to as what?
-
Which calculation would reveal the bias in a forecasting technique, the mean error or mean absolute deviation?
-
When gasoline is refined from crude oil, each gallon of regular, mid-grade, and premium uses a different blend of two different grades of crude oil according to the following matrix. In addition, the...
-
Vectors are drawn from the center of a regular n-sided polygon in the plane to the vertices of the polygon. Show that the sum of the vectors is zero.
-
A compound of cobalt, carbon, and oxygen contains 28.10% C and 34.47% Co. When 0.147 g of this compound is dissolved in 6.72 g of cyclohexane, the solution freezes at 5.23C. What is the molecular...
-
The carbohydrate digitoxose contains 48.64% carbon and 8.16% hydrogen. The addition of 18.0 g of this compound to 100. g of water gives a solution that has a freezing point of -2.2C. a. What is the...
-
Analysis of a compound gave 39.50% C, 2.21% H, and 58.30% Cl. When 0.855 g of this solid was dissolved in 7.50 g of naphthalene, the solution had a freezing point of 78.0C. The pure solvent freezes...
-
What is one of the differences between prime costs and conversion costs in manufacturing? A) Only conversion costs include direct labor. B) Only prime costs include manufacturing overhead. Only...
-
MTN H+ X 2024 rt 1 of 1 - 100% 6:46 sakai.ug.edu.gh/porta 73 Question 26 of 40 The basis of the management control function is A. decision making. B. planning. OC. organizing. D. accounting...
-
An organization is planning their master budget. They begin by listing all of their expected cash outflows, including employee salaries and benefits, utilities, program expenses, insurance, vehicle...

Study smarter with the SolutionInn App


