The element X below could be a. Sulfur b. Iodine c. Aluminum d. Phosphorus e. Silicon X.
Question:
The element X below could be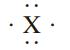
a. Sulfur
b. Iodine
c. Aluminum
d. Phosphorus
e. Silicon
Transcribed Image Text:
X.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
The ...View the full answer

Answered By

Amos Kiprotich
I am a wild researcher and I guarantee you a well written paper that is plagiarism free. I am a good time manager and hence you are assured that your paper will always be delivered a head of time. My services are cheap and the prices include a series of revisions, free referencing and formatting.
4.90+
15+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The thin outer layer of Earth, called the crust, contains only 0.50 percent of Earth's total mass and yet is the source of almost all the elements (the atmosphere provides elements such as oxygen,...
-
Bea Jones (age 32) moved from Texas to Florida in December 2013. She lives at 654 Ocean Way, Gulfport, FL 33707. Beas Social Security number is 466787359 and she is single. Her earnings and income...
-
Silicon is just below carbon in the periodic table. Predict the geometry of silicon tetrafluoride, SiF4.
-
In Exercises 7683, use a graphing utility to graph the function. Use the graph to determine whether the function has an inverse that is a function (that is, whether the function is one-to-one). f(x)...
-
On September 12, the cheapest-to-deliver bond on the December Treasury bond futures contract is the 9s of November YY+18. The bond pays interest semiannually on May 15 and November 15. Its price is...
-
Full-Capacity Sales Seaweed Mfg., Inc., is currently operating at only 90 percent of fixed asset capacity. Current sales are $610,000. How fast can sales grow before any new fixed assets are needed?
-
The following information for Great Oaks Furniture, a retail furniture and design firm, relates to Exercises 6 through 11. Perform a horizontal analysis of the balance sheets and income statements...
-
Miyamoto Jewelers is considering a special order for 10 handcrafted gold bracelets to be given as gifts to members of a wedding party. The normal selling price of a gold bracelet is $389.95 and its...
-
Teamworks - Home Welcome to Ashwo. E Home | LDS Busines... Online University-.. M Inbox (20,736) - sj8... G Google (116) YouTube Al Paraphrasing To... Favorites Other favorit Home Client Profile...
-
In the fiscal year that ended September 30, 2008, there were 24,584 age-discrimination claims filed with the Equal Employment Opportunity Commission, an increase of 29% from the previous year (The...
-
Which of the following represent configurations of indium ions in compounds? Explain your decision in each case. a. In 4+ [Kr]4d 9 b. In + [Kr]4d 10 5s 2 c. In 3+ [Kr]4d 10 d. In [Kr]4d 10 5s 1
-
Which of the following is the ground-state electron configuration of a C 3- ion? a. 1s 2 2s 2 2p 4 b. [He]2s 2 2p 6 c. [He]2s 1 d. 1s 2 2s 2 2p 5 e. 1s 2 2s 2 2p 6 3s 1
-
Draw the general shape of the temperatureenergy graph for the heating of ethanol from -120 C to 120 C. (Given: Mp = -117.3 C; Bp = 78.5 C).
-
Imagine for a moment a relaxing day at the beach, when suddenly you hear the sound of someone in the ocean who appears to be in distress. Next, picture the arrival of a drone, an unmanned aerial...
-
A military base in the United States is examining the distribution of the amount of time that officers on the base have been in the military. The following data show the years of military service for...
-
List the four principles that underpin the SEC's auditor independence rules.
-
List the restrictions placed on auditors by the professional accountancy bodies which are designed to prevent auditors' independence from being impaired through: (i) financial involvement with an...
-
Describe briefly the changes to auditors' responsibilities in respect of detecting fraud: (i) introduced in (revised) ISA 240: Fraud and error; (ii) indicated in the APB's Consultative Papers: Fraud...
-
Ace, Baker, and Champ are being purchased by ALCO, Inc., and their outstanding debts must be paid by the purchaser. The following matrix gives the amounts of debt and the terms for the companies...
-
In Exercises 516, find the focus and directrix of the parabola with the given equation. Then graph the parabola. y 2 = 4x
-
An antiseptic solution contains hydrogen peroxide, H2O2, in water. The solution is 0.600 m H2O2. What is the mole fraction of hydrogen peroxide?
-
Concentrated hydrochloric acid contains 1.00 mol HCl dissolved in 3.31 mol H2O. What is the mole fraction of HCl in concentrated hydrochloric acid? What is the molal concentration of HCl?
-
Concentrated aqueous ammonia contains 1.00 mol NH3 dissolved in 2.44 mol H2O. What is the mole fraction of NH3 in concentrated aqueous ammonia? What is the molal concentration of NH3?
-
AC 0 ezto.mheducation.com 13e_ch15_final student - Fa23 SAMG 1200-22... G11. What is a 'proper promotional mix' - Google Sea... Chapter 14 Homework by Sunday, 11/12 i M Question 8-Chapter 14 Homework...
-
23 BUSM 1260-22 Applied Business Mathematics/Calculators ezto.mheducation.com M Question 1- Chapter 14 Homework by Sunday, 11/12 - Connect 4 Homework by Sunday, 11/12 ok nt rences Complete the...
-
ABC Bank has assets of $120 and a leverage ratio of 3. It earns 5% on its assets but pays 4% on its liabilities. What is ABC Bank's profit per unit of capital invested? X (round to the nearest...

Study smarter with the SolutionInn App


