What is G at 25C for the reaction Sn 2+ (aq) + 2Hg 2+ (aq) Sn
Question:
What is ∆G° at 25°C for the reaction
Sn2+(aq) + 2Hg2+(aq) → Sn4+(aq) + Hg22+(aq)
For data, see Table 19.1.
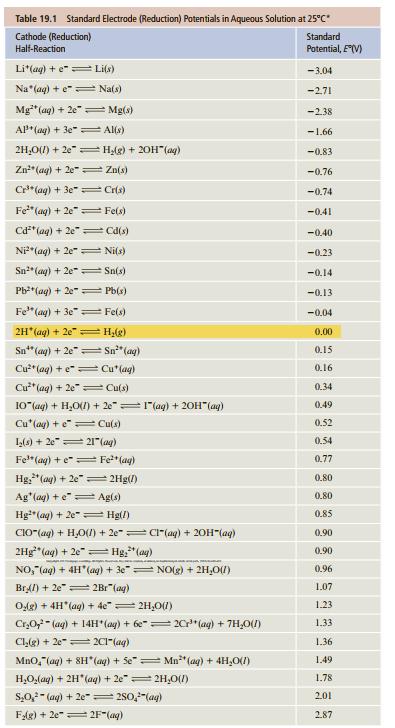
Transcribed Image Text:
Table 19.1 Standard Electrode (Reduction) Potentials in Aqueous Solution at 25°C Standard Cathode (Reduction) Half-Reaction Potential, E(V) Li*(ag) + e"= Li(s) - 3.04 Na"(aq) + e"= Na(s) -2.71 Mg"(ag) + 2e" = Mg(s) -2.38 AP* (ag) + 3e" = Al(s) -1.66 2H,O(1) + 2e"= Hlg) + 20H"(ag) -0.83 Zn"(ag) + 2e = Zn(s) -0.76 Cr*(aq) + 3e" Cris) -0.74 Fe" (ag) + 2e" Fe(s) -0.41 Cd*(ag) + 2e" = Cd(s) -0.40 Ni*(aq) + 2e" = Ni(s) -0.23 Sn"(ag) + 2e Sn(s) -0.14 Pb*(ag) + 2e" = Pb(s) -0.13 Fe" (ag) + 3e" = Fe(s) -0.04 2H (ag) + 2e H,(g) 0.00 Sn" (ag) + 2e"= Sn" (ag) 0.15 Cu**(aq) + e"= Cu*(ag) Cu**(ag) + 2e"= Cu(s) 10"(ag) + H,O) + 20"=1"(ag) + 20H" (ag) 0.16 0.34 0.49 Cu'(ag) + e= Cu(s) 0.52 0.54 L(s) + 2e"= 21"(ag) Fe"(ag) + e= Fe*(ag) 0.77 Hg;" (ag) + 2e =2Hg(/) 0.80 Ag'(ag) + e" Ag(s) 0.80 Hg"(ag) + 2e" = Hg(/) 0.85 CIO"(ag) + H,O() + 2e"= CI"(aq) + 20H"(ag) 2Hg" (aq) + 2e" = Hg," (ag) NO," (ag) + 4H (ag) + 3e"= NO(g) + 2H,01) 0.90 0.90 0.96 Brl) + 2e" 2Br"(ag) 1.07 Odg) + 4H"(ag) + 4e"= 2H,0(7) 1.23 Cr,O,- (ag) + 14H (ag) + 6e"= 2C"(ag) + 7H,0(1) 1.33 Cl,g) + 2e"= 2C"(ag) 1.36 Mno,"(aq) + SH (aq) + Se" Mn** (ag) + 4H,0(1) 1.49 H,O(ag) + 2H"(aq) + 2e"= 2H,0(1) 1.78 S,0,- (ag) + 2e-=280,"(ag) 2.01 FAg) + 2e" 2F"(ag) 287
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
The halfcell reactions the corresponding halfcell potentials and their sums are displayed ...View the full answer

Answered By

WAHIDUL HAQUE
hello,
I'm a professional academic solution provider working as a freelance academic solution provider since 7 years. I have completed numerous projects. Help lots of students to get good marks in their exams and quizzes. I can provide any type of academic help to your homework, classwork etc, if you are a student of Accounting, Finance, Economics, Statistics. I believe in satisfying client by my work quality, rather than making one-time profit. I charge reasonable so that we make good long term relationship. why will you choose me? i am an extremely passionate, boldly honest, ethically driven and pro-active contractor that holds each of my clients in high regards throughout all my business relations. in addition, I'll always make sure that I'm giving my 100% better in every work that will be entrusted to me to be able to produce an outcome that will meet my client's standards. so if you are a student that is now reading my profile and considering me for your academic help. please feel free to look through my working history, feedback and contact me if you see or read something that interests you. I appreciate your time and consideration.
regards
4.90+
233+ Reviews
368+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Table 10.16 gives data on the crime rate in 47 states in the United States for 1960. Try to develop a suitable model to explain the crime rate in relation to the 14 socioeconomic variables given in...
-
Attached are two data sets, one concerning Skittles and one about cereals. You may assume both samples provided were selected randomly and are representative of the population. Be sure to use the...
-
In Example 15.6, pure-component, liquid-phase adsorption data are used with the extended-Langmuir isotherm to predict a binary-solute data point. Use the following mixture data to obtain the best fit...
-
Find the volumes of the solids in Problems 4952 correct to the nearest unit. 4 cm -6 cm 8 cm
-
One of the most difficult tasks of developing and managing a global portfolio is assessing the risks of potential foreign investments. Duke University researcher C. R. Henry collaborated with two...
-
Revenue and production budgets. (CPA, adapted) The Scarborough Corporation manufactures and sells two products: Thingone and Thingtwo. In July 2009, Scarboroughs budget department gathered the...
-
\(1,345,321\) Use divisibility rules to determine if each of the following is divisible by 5 .
-
You have been given a file that contains fields relating to CD information. Using the steps of normalization, create a logical data model that represents this file in third normal form. The fields...
-
Please provide any formulas used to get to the answer. 4. Fashion Trends, Inc., a regional fashion apparel retailer, wants to prepare a 2018 Pro Forma Income Statement and a 2018 Balance Sheet using...
-
Write a line of code that performs each of the following tasks: a) Specify that class PieceWorker inherits from class Employee. a) Call superclass Employees toString method from subclass PieceWorkers...
-
Define the faraday.
-
Consider the reducing agents Sn 2+ (aq), Cl 2 (g), and I (aq). Which is strongest? Which is weakest?
-
Determine the following indefinite integrals. dx 16 + x
-
When multiple-choice questions are asked, the order of the choices is usually changed each time the question is asked. For example, in the 2016 presidential election, a pollster would ask one person...
-
In the 1936 presidential election, Republican candidate Alf Landon challenged President Franklin Roosevelt. The Literary Digest magazine conducted a poll in which they mailed questionnaires to more...
-
A college faculty consists of 400 men and 250 women. The college administration wants to draw a sample of 65 faculty members to ask their opinion about a new parking fee. They draw a simple random...
-
A Queensland Industrial Relations Commission case in 2018 highlighted the fact that everybody loses when racial discrimination enters the workplace.44 Two former friends fell out over a series of...
-
Find an example of an undirected network in the real world. Provide justi-fication as to why this network operates as an undirected network.
-
Perform the indicated operations with expressions involving fractional exponents and radicals and then simplify. 1. x1/2 (x1/2 + 2x3/2) 2. x-2/3(x5/3 - x-1/3) 3. (x1/2 + 1) (x1/2 - 2) 4. (x1/3 -...
-
You work as an operations consultant for a textile company. Your client has a well-established distribution system in the US market. The company has hundreds of stores and four distribution centers....
-
Formic acid, HCHO2, is a stronger acid than acetic acid, HC2H3O2. Which is the stronger base, formate ion, CHO2, or acetate ion, C2H3O2?
-
Rank the following solutions from most acidic to most basic (water molecules have been omitted for clarity).
-
You have solutions of NH3, HCl, NaOH, and HC2H3O2 (acetic acid), all with the same solute concentrations. Rank these solutions in order of pH, from the highest to the lowest.
-
Joaquin is using the survey method of a job analysis for a barista position at Peet's Coffee. Ideally Joaquin should give the survey to Group of answer choices a secret shopper a customer a position...
-
In 2 0 1 9 Illinois passes a law which will require inspection of any public places to determine if the paint used on the walls contains lead. Illinois will charge the paint manufacturer of the paint...
-
A job interview that involves one job applicant and two or more interviewers is called Group of answer choices an applied interview a multiple group setting interview structured interview a panel...

Study smarter with the SolutionInn App


