With the aid of a periodic table (not Figure 9.15), arrange the following in order of increasing
Question:
With the aid of a periodic table (not Figure 9.15), arrange the following in order of increasing electronegativity:
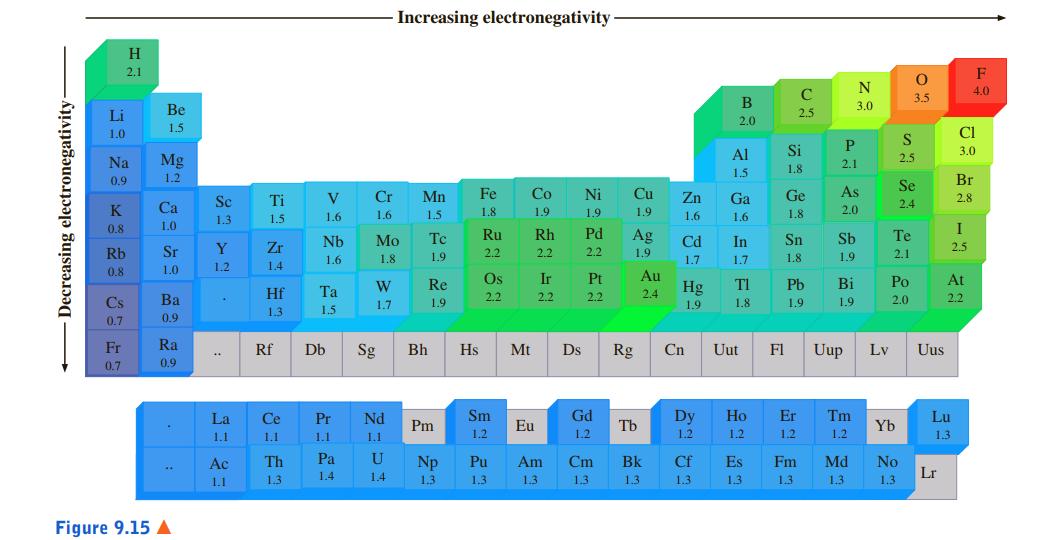
a. Li, Na, Cs
b. B, Be, Li
c. S, Se, Cl
Transcribed Image Text:
Increasing electronegativity H 2.1 F N 4.0 C 3.5 B 3.0 Li Be 2.5 2.0 1.5 1.0 CI Al Si 3.0 Na Mg 2.1 2.5 1.5 1.8 0.9 1.2 Br Se V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Ti 2.4 2.8 K Ca 1.5 2.0 1.6 1.6 1.5 1.8 1.9 1.9 1.9 1.6 1.6 1.8 1.3 1.5 0.8 1.0 I Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te Sr Y Zr 2.5 Rb 1.8 1.9 2.2 2.2 2.2 1.9 1.8 1.9 2.1 1.6 1.7 1.7 0.8 1.0 1.2 1.4 Re Os Ir Pt Au TI Pb Bi Po At Hf Ta Hg Cs Ba 2.2 2.2 2.2 2.4 1.9 2.0 2.2 1.7 1.9 1.9 1.8 1.9 1.3 1.5 0.7 0.9 Fr Ra Rf Db Sg Bh Hs Mt Ds Rg Cn Uut F1 Uup Lv Uus 0.7 0.9 La Ce Pr Nd Sm Gd Dy Ho Er Tm Lu Pm Eu Tb Yb 1.1 1.1 1.1 1.1 1.2 1.2 1.2 1.2 1.2 1.2 1.3 Ac Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No 1.4 1.4 Lr 1.1 1.3 1.3 1,3 1.3 1.3 1.3 1.3 1.3 1.3 1.3 1.3 Figure 9.15 A Decreasing electronegativity Wn
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
a Cs Na Li Electronegativity increases from bottom to ...View the full answer

Answered By

Sumit kumar
Education details:
QUATERNARY Pursuing M.Tech.(2017-2019) in Electronics and Communication Engg. (VLSI DESIGN) from
GNIOT Greater Noida
TERTIARY B.Tech. (2012-2016) in Electronics and Communication Engg. from GLBITM Greater Noida
SECONDARY Senior Secondary School Examination (Class XII) in 2012 from R.S.S.Inter College, Noida
ELEMENTARY Secondary School Examination (Class X) in 2010 from New R.J.C. Public School ,Noida
CERTIFICATION
Summer Training in ‘WIRELESS EMBEDDED SYSTEM’ from ‘XIONEE’ for the six weeks.
EMBEDDED SYSTEM Certificate issued by CETPA INFOTECH for one day workshop.
Certificate of Faculty development program on OPTICAL COMMUNICATION and NETWORKS for one week.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Arrange the following in order of increasing basicity: (a) (b) (c)
-
Arrange the following in order of increasing first ionization energy: Na, Cl, Al, S, and Cs.
-
Arrange the following in order of increasing first ionization energy: F, K, P, Ca, and Ne.
-
In Exercises 1 through 28, differentiate the given function. y = 2x
-
On July 1, a portfolio manager holds $1 million face value of Treasury bonds, the 11 l/4s maturing in about 29 years. The price is 107 14/32. The bond will need to be sold on August 30. The manager...
-
Peer Group Analysis Explain what peer group analysis is. As a financial manager, how could you use the results of peer group analysis to evaluate the performance of your firm? How is a peer group...
-
To examine how much cash your firm has returned to its stockholders and in what form (dividends or stock buybacks) and to evaluate whether the trade-off favors returning more or less. Key Questions ...
-
Fernandez Corporation purchased a truck at the beginning of 2012 for $50,000. The truck is estimated to have a salvage value of $2,000 and a useful life of 160,000 miles. It was driven 23,000 miles...
-
how did we get 36 million?. Scepter lost a lot of money in purchasing ATH Terminal value = CF/ (r - g ) = $36million/(0.18 - 0.1) = $450million G = growth rate) Good Investment >$184million paid by...
-
Consider the following information about a university database: Professors have an SSN, a name, an age, a rank, and a research specialty. Projects have a project number, a sponsor name (e.g., NSF),...
-
Decide which of the following bonds is least polar on the basis of electronegativities of atoms: ClH, SSi, BrAs.
-
Assuming that the atoms form the normal number of covalent bonds, give the molecular formula of the simplest compound of germanium and fluorine atoms.
-
What is the purpose of organizational design?
-
Define and explain specific fuel consumption.
-
Give the merits and demerits of four-stroke and two-stroke cycle engines.
-
Define the following : (a) Indicated power (b) Brake power (c) Mechanical efficiency (d) Indicated and brake thermal efficiency (e) Relative efficiency
-
List applications of I.C. engines.
-
Why petrol engines are called S.I. engines and why diesel engines are called C.I. engines.
-
Use technology to find the product AB of the following matrices. 1. 2. 0.1 0.1 0.0 0.1 0.2 0.1 0.2 -0.1 0.1 -0.1 A = 0.1 -0.1 0.1 -0.2 0.1 0.1 0.1 0.2 0.1 0.1 0.1 0.0 0.0 0.0 0.0 10 10 5 -10 20 - 10...
-
Use translations to graph f. f(x) = x-/2 +1
-
A solution is made up by dissolving 15.0 g Na2CO310H2O in 100.0 g of water. What is the molality of Na2CO3 in this solution?
-
An aqueous solution is 15.0% by mass of copper(II) sulfate pentahydrate, CuSO45H2O. What is the molarity of CuSO4 in this solution at 20C? The density of this solution at 20C is 1.167 g/mL.
-
An aqueous solution is 20.0% by mass of sodium thiosulfate pentahydrate, Na2S2O35H2O. What is the molarity of Na2S2O3 in this solution at 20C? The density of this solution at 20C is 1.174 g/mL.
-
Sample size in physical observation of inventory is Question content area bottom Part 1 A. determined using attributes sampling. B. difficult to specify because the emphasis is on observing the...
-
If we receive a refund from our federal income tax, what does this mean? A. The right amount of withholding was taken from our pay the previous year. B. Too much tax was withheld from our pay the...
-
Question 12 4 Points Great North Waters produces the highest quality bottled water in the country. The company purchases water from a local quarry, stores the water in large tanks, and then runs the...

Study smarter with the SolutionInn App


