Substitution of H 2 O in [Fe(OH 2 ) 6 ] 3+ by thiocyanate is complicated by
Question:
Substitution of H2O in [Fe(OH2)6]3+ by thiocyanate is complicated by proton loss. By considering the reaction scheme below, derive an expression for
![]()
in terms of the equilibrium and rate constants, [Fe(OH2)63+], [SCN]−, [Fe(OH2)5(SCN)2+] and [H+].
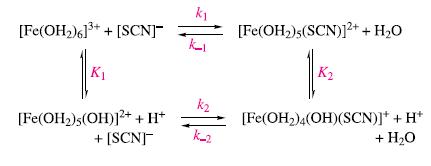
Transcribed Image Text:
d[SCN] dt
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To derive an expression for the rate of the reaction involving the substitution of H2O in FeOH263 by thiocyanate SCN in the presence of protons H we n...View the full answer

Answered By

Mary Boke
I have teached the student upto class 12th as well as my fellow mates.I have a good command in engineering,maths and science.I scored 90+ marks in 10th and 12th in maths.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The net potential energy EN between two adjacent ions is sometimes represented by the expression in which r is the interionic separation and C, D, and Ï are constants whose values depend on the...
-
You start driving east for 14 miles, turn left, and drive north for another 10 miles. At the end of driving, what is your straight line distance from your starting point? Round to the nearest tenth...
-
Flowers Associates is evaluating the performance of three divisions: Daisies, Pansies, and Tulips. Using the data that follow, compute the return on investment and residual income for each division,...
-
A spherical particle with $\mathrm{SG}=2.5$ and a diameter of $2 \mathrm{~mm}$ is immersed in water in a cylindrical centrifuge with a diameter of $20 \mathrm{~cm}$. If the particle is initially $8...
-
An investor purchased 100 shares of Mallard common stock at \(\$ 20\) per share on March 15, 2020. On December 31, 2020, the stock was quoted at \(\$ 19\) per share and declared and paid a dividend...
-
1. Trace the path of the medication list and denote possible failure points. Construct a process flowchart of the existing process and create a new chart of an improved process. 2. Was the medication...
-
On May 1, 2024, Pharoah Corp. issued $648,000 of 9%, 5-year unsecured bonds at face value. The bonds were dated May 1, 2024, and pay interest annually on May 1. Financial statements are prepared...
-
Rationalize the observation that when the reaction: is carried out in H 2 ( 18 O), the water in the complex contains equal proportions of H 2 ( 18 O) and H 2 ( 16 O). [Co(NH3)4 (CO3)]* [HO]*, HO 13+...
-
(a) Rationalize the formation of the products in the following sequence of reactions: (b) Suggest methods of preparing [RhCl 5 (OH 2 )] 2 , cis-[RhCl 4 (OH 2 ) 2 ] and fac-[RhCl 3 (OH 2 ) 3 ]....
-
International Manufacturing decides to acquire a small local manufacturing company called Township Manufacturing. Township Manufacturing has assets with a market value of $120,000 and no liabilities,...
-
Simplify. 2w9v74w8 x 9.2xv7 -7 Use only positive exponents in your answer.
-
Do you believe that your present duties and work are in line with your long-term objectives?
-
1. What are some specific factors that make countries unattractive to firms looking to do business there? 2. What are some of the factors that firms must consider before deciding to begin...
-
a) Perform Girvan Newman Algorithm on the following network to find hierarchical communities. Show each step. 1.00 0.75 0.50 0.25 0.00 -0.25 -0.50 -0.75 -1.00 -0.8 -0.6 -0.4 -0.2 0.0 02 04 0.6 b)...
-
A plane traveling north at 120 knots passes directly over thePrescott airport. A second plane passes east over the airport onehour later, at a speed of 100 knots. Both planes are at the samealtitude....
-
The 10 cars in Table 28 have been randomly selected for the sample of an experiment. Use the seed 469 to randomly select 5 of the cars for the treatment group. The other 5 cars will be in the control...
-
Independent random samples of sizes n1 = 30 and n2 = 50 are taken from two normal populations having the means 1 = 78 and 2 = 75 and the variances 21 = 150 and 22 = 200. Use the results of Exercise...
-
Draw a plausible mechanism for the following transformation: , [H2SO4]
-
Compound A has molecular formula C 8 H 14 O 2 . Upon treatment with catalytic acid, compound. A is converted into the cyclic hemiacetal. Identify the structure of compound A. , Compound A [H*]
-
Draw a plausible mechanism for each of the following transformations: (a) (b) [TSOH] MENH2 -H20 Et [TSOH] EENH2 -H20
-
A company sellis in a competitive market and currently noticed a decline in sales. It sells its product for $300. The market price for the product is $270 and the target return on sales is 15%. The...
-
Company provided the following cost formulas for three of its expenses (where q refers to the number of hours worked): Rent (fixed) Supplies (variable) Utilities (mixed) $ 3,000 $ 4.00g $150 +$0.75g...
-
You are about to optimize the IP addressing table for a network with 5 different subnetworks and 20 hosts pers subnetwork. The Internet Service Provider gives you a class B network. The best...

Study smarter with the SolutionInn App


