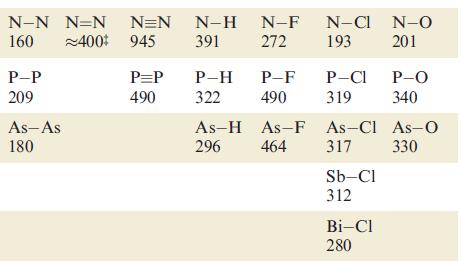
Using bond enthalpy terms from Tables 14.2 and 15.3, estimate values of r H for the
Question:
Using bond enthalpy terms from Tables 14.2 and 15.3, estimate values of ΔrHº for the following reactions
(a) 2N2 → N4 (tetrahedral structure);
(b) 2P2 → P4 (tetrahedral structure);
(c) 2C2H2 → C4H4 (tetrahedrane, with a tetrahedral C4 core).
Table 14.2.
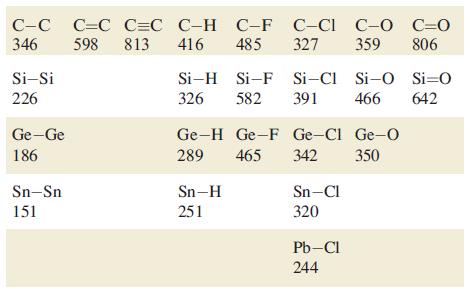
Table 15.3.
Transcribed Image Text:
C-C_C=CC=C_C-H C-F C-CI C-0_C=0 598 813 416 485 327 359 806 346 Si-Si 226 Ge-Ge 186 Sn-Sn 151 Si-H Si-F Si-Cl Si-O Si=0 582 391 466 326 642 Ge-H Ge-F 289 465 342 350 Ge-Cl Ge-0 Sn-H 251 Sn-Cl 320 Pb-Cl 244
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To estimate the values of rH standard enthalpy change for the given reactions using bond enthalpy te...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using data from Appendix 4, calculate ÎHo, ÎSo, and DGo for the following reactions that produce acetic acid: Which reaction would you choose as a commercial method for producing acetic...
-
For the following reactions at constant pressure, predict if H . E, H , E, or H = E. a. 2HF(g) H2(g) + F2(g) b. N2(g) + 3H2(g) 2NH3(g) c. 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(g)
-
Using the data in Appendix 3, calculate the standard entropy changes for the following reactions at 25C: (a) S(s) + O2(g) SO2(g) (b) MgCO3(s) MgO(s) + CO2(g)
-
Consider each of the following situations. Indicate whether (and why or why not) you think that the governement should account for the transactions and resources in an agency fund, a governmental...
-
A number of business transactions carried out by Smalling Manufacturing Company are as follows: a. Borrowed money from a bank. b. Sold land for cash at a price equal to its cost. c. Paid a liability....
-
In the interference pattern created by light diffracted from a single slit, which are wider: the first-order bright fringes or the third-order bright fringes? (Plot or plug in typical numerical...
-
Gomez Inc. leases a vehicle from CareMax Inc. on January 1, 2020, for a three-year period, appropriately classified by Gomez Inc. as an operating lease. Gomez agrees to make \(\$ 6,000\) annual...
-
Lafayette Film Center (LFC) is a not-for-profit theater that plays independent films. In addition to revenue from theater admissions, LFC relies on concession and caf sales, grants and other external...
-
The ledger of Oriole Company contains the following balances: Owner's Capital $ 29,300, Owner's Drawings $ 3,000, Service Revenue $ 50,000, Salaries and Wages Expense $ 26,000, and Supplies Expe...
-
Lead-acid batteries accounted for 69% of all lead consumed in the US in 2015. (a) Complete the cell reaction given below (not balanced on the left-hand side) and show that the oxidation state changes...
-
The glass industry manufactures millions of tonnes of glass per year. (a) Only certain element oxides form glasses. Explain why this is, giving examples of what are termed in the glass industry as...
-
For Example 1 in this section, the hypothesis test has power of 0.4274 of supporting the claim that < 1.00 W/kg when the actual population mean is 0.80 W/kg. a. Interpret the given value of the...
-
Describe 5 global design-innovation trends that apply to telecommunications network engineering?
-
What common marketing strategies are considered illegal in the US? What common marketing strategies are illegal overseas?
-
2) Convert to PIC24 assembly: uint8 u8_i,u8_j,u8_k; if ((u8 j & 0x01) == 1) { u8_k++; } u8_ju8_j 1 u8_i++; }
-
What is the difference between the Network Load Balancer and the Application Load Balancer? Provide an example on how an organization uses the application Load Balancer.
-
You were testing 300 units of a product using accelerated life testing equivalent to 2,000 hours of normal use. One of your units failed after 500 hours, another after 1,000 hours, and another after...
-
Determine which of the following 2014 annual gifts are subject to gift taxes and to what extent they need to be included in an estate. a. Grandparents gave a grandchild $24,000 for the purchase of a...
-
The National Collegiate Athletic Association (NCAA) and the National Federation of State High School Associations (NFHS) set a new standard for non-wood baseball bats. Their goal was to ensure that...
-
Draw the B 12 unit that is a common motif of boron structures; take a viewpoint along a C 2 axis.
-
Give balanced chemical equations for the synthesis of 1,2-B 10 C 2 H 10 (Si(CH 3 ) 3 ) 2 starting with decaborane(14) and other reagents of your choice.
-
Give the IUPAC names of (a) B 10 H 14 , (b) [B 12 H 12 ] 2 , (c) Arachno-[B 12 H 14 ] 2 .
-
The largest portion of budgeted project costs come from: Question 1 0 options: Management Materials Indirect costs Resources
-
The higher the DOL, the _ _ _ _ _ _ _ _ _ _ change you will see in operating income with a change in sales.
-
Audit Procedures for Financial Instruments and Derivatives: Case Study: Alpha Bank, a leading financial institution, engages in various activities involving financial instruments and derivatives. As...

Study smarter with the SolutionInn App


