Consider a chloroform(1)/tetrahydrofuran(2) mixture with x 1 = 0.90, initially at 76C and 120 kPa. Describe the
Question:
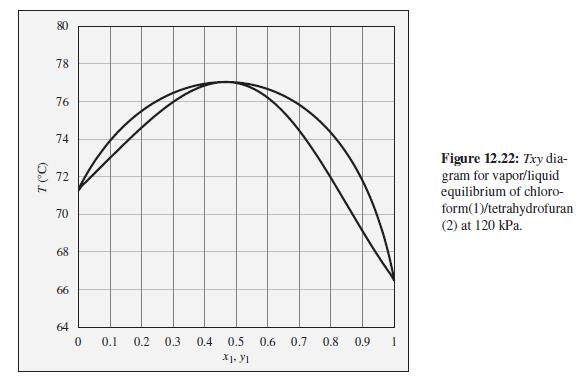
Consider a chloroform(1)/tetrahydrofuran(2) mixture with x1 = 0.90, initially at 76°C and 120 kPa. Describe the evolution of phases and phase compositions as the temperature is gradually reduced to 66°C.
To the Txy diagram for chloroform(1)/tetrahydrofuran(2) at 120 kPa shown in Fig. 12.22.
Transcribed Image Text:
78 76 74 Figure 12.22: Txy dia- gram for vapor/liquid equilibrium of chloro- form(1)/tetrahydrofuran (2) at 120 kPa. 72 70 68 66 64 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 X1. Y1 T (C)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
As the temperature of the chloroform1tetrahydrofuran2 mixture is gradually reduced from 76C to 66C i...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
Describe the evolution of business-to-business (B2B) e-commerce.
-
Describe the evolution of ERP systems.
-
Describe the evolution of American culinary arts.
-
Greety Food in Ashland, Kentucky, manufactures and markets snack foods. Sita Lee manages the company's fleet of 220 delivery trucks. Lee has been charged with *reengineering* the fleet-management...
-
Whatever can be done by a tariff can be done by a quota. Discuss.
-
What is the fixed overhead spending variance? Who is most likely responsible for it?
-
The following information from Buchanan Company's current operations is available: Required a. Prepare a multiple-step income statement. Disregard earnings per share. b. Prepare a single-step income...
-
At the start of the current year, Blue Corporation (a calendar year taxpayer) has accumulated E & P of $100,000. Blue's current E & P is $60,000, and at the end of the year, it distributes $200,000...
-
Job number 2288 had the following data: Direct Labor hours used 238 Supplies used $450 Hourly wage rate $20 per hour Overhead allocation rate $8 per direct labor hour Assuming this was the only job...
-
The Gaubert Marketing Company needs the following number of telemarketers on the phones during the upcoming week: Monday 23, Tuesday 16, Wednesday 21, Thursday 17, Friday 20, Saturday 12, and Sunday...
-
A certain gas is described by the equation of state: Here, b is a constant and is a function of T only. For this gas, determine expressions for the isothermal compressibility and the thermal...
-
Consider a chloroform(1)/tetrahydrofuran(2) mixture with x 1 = 0.10, initially at 80C and 120 kPa. Describe the evolution of phases and phase compositions as the temperature is gradually reduced to...
-
Use a change of variables to evaluate the following integrals. x dV; D is bounded by the planes y - 2x = 0, y 2x = 1, D z - 3y = 0, z - 3y = 1,2 - 4x = 0, and z 4x = 3.
-
Early in 2002, WorldCom suffered a decline in business as a result of: (a) The decline of acquisitions to falsify profits. (b) The fall of stock price. (c) Ebbers loans, which were eating cash. (d)...
-
If a company overstates the reserves of a company that it acquires and increases goodwill by a corresponding amount in the period of the acquisition in order to release the reserves in a later...
-
At Enron Energy Services, many of the contracts were overvalued, as the energy loads required by its customers were based on guesswork and excessively optimistic assumptions. True/False
-
WorldCom improperly accounted for its line cost expense by: (a) Allocating line-cost expense to PPE in its first recording of the line-cost transactions. (b) Initially properly debiting the line-cost...
-
If a parent company discovers that the assets of an acquired company are worth less than it believed at the time of the acquisition, it is appropriate to increase goodwill because this means that the...
-
James Ltd.'s statement of fi nancial position at December 31, 2016, is presented below. During 2017, the following transactions occurred. 1. James paid £2,500 interest on the bonds on January...
-
The Alert Company is a closely held investment-services group that has been very successful over the past five years, consistently providing most members of the top management group with 50% bonuses....
-
A two-stage cascade refrigeration system (see Fig. 93) operates between TC = 210 K and TH = 305 K. Intermediate temperatures are T'C = 255 K and T'H = 260 K. Coefficients of performance of each...
-
A two-stage cascade refrigeration system (see Fig. 93) operates between TC = 210 K and TH = 305 K. Intermediate temperatures are T'C = 255 K and T'H = 260 K. Coefficients of performance of each...
-
The contents of the freezer in a home refrigerator are maintained at - 20C. The kitchen temperature is 20C. If heal leaks amount to 125.000 U per day and if electricity costs $0.08/k Whr, estimate...
-
Explain the role of performance measurement in managing supply chain activities. Macec
-
1- Explain how the analytical tools for Six Sigma and continuous improvement can be applied to assure the dimensions of design quality are achieved relative to supply chain management. Link this...
-
If the HIV Ab test came back negative, what is the probability that the patient does not have HIV infection? HIV Ab test information: sensitivity = 90% specificity = 90% positive predictive value =...

Study smarter with the SolutionInn App


