The pressure above a mixture of ethanol and ethyl acetate at 70C is measured to be 78
Question:
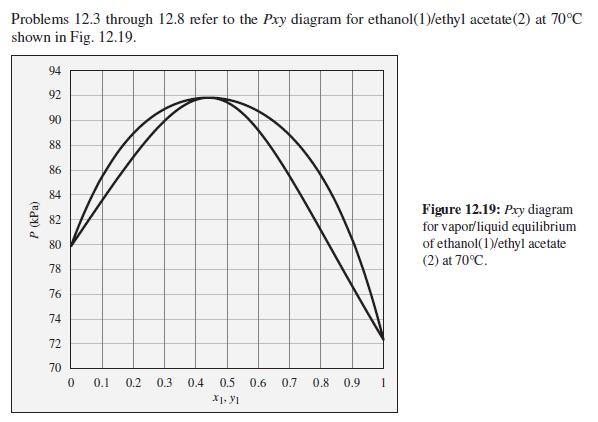
The pressure above a mixture of ethanol and ethyl acetate at 70°C is measured to be 78 kPa. What are the possible compositions of the liquid and vapor phases?
Transcribed Image Text:
Problems 12.3 through 12.8 refer to the Pxy diagram for ethanol(1)/ethyl acetate(2) at 70°C shown in Fig. 12.19. 94 92 90 88 86 84 Figure 12.19: Pry diagram for vapor/liquid equilibrium of ethanol(1)/ethyl acetate (2) at 70°C. 82 80 78 76 74 72 70 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 X1. y1 P (kPa)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
A relativistic rocket is measured to be 50 m long, 2.5 m high, and 2.0 m wide by its pilot. It is traveling at 0.65c (in the direction parallel to its length) relative to an inertial observer. (a)...
-
A mixture of ethanol and 1-propanol behaves ideally at 36C and is in equilibrium with its vapor. If the mole fraction of ethanol in the solution is 0.62, calculate its mole fraction in the vapor...
-
The pressure of an automobile tire is measured to be 190 kPa (gage) before a trip and 215 kPa (gage) after the trip at a location where the atmospheric pressure is 95 kPa. If the temperature of air...
-
Count Dracula, the most famous vampire, rumored to have killed at least 200,000 people, was based on a real person who lived in eastern Europe about 600 years ago. He was indeed a "monster," although...
-
Is it possible for everyones real income to rise even though the income distribution in a society has become more unequal? Prove your answer with a numerical example.
-
Why can the current movement toward establishing supply chains be characterized as a revolution?
-
How much do we need to invest each year to have \($100,000.00\) in a college fund when our child turns 18 years old? New parents often seek similar advice from their financial advisors to establish...
-
In 2014 Marquette County opened a landll that was expected to accept waste for four years. The following table indicates the estimates county ofcials made at the end of each of the four years:...
-
If you borrow $100,000 at 7% fixed interest for 30 years in order to buy a house, what will be your monthly house payment? After graduation, you plan to invest $400 per month in the stock market.If...
-
1. Gugenheim, Inc. offers a 12 percent coupon bond with annual payments. The yield to maturity is 5.60 percent and the maturity date is 11 years. What is the market price of a $1,000 face value bond?...
-
Consider an ethanol(1)/ethyl acetate(2) mixture with x 1 = 0.70, initially at 70C and 100 kPa. Describe the evolution of phases and phase compositions as the pressure is gradually reduced to 70 kPa....
-
Of the following binary liquid/vapor systems, which can be approximately modeled by Raoults law? For those that cannot, why not? Table B.1 (App. B) may be useful. (a) Benzene/toluene at 1(atm). (b)...
-
Lenders tend to offer lower interest rates to borrowers with high credit scores and higher interest rates to borrowers with low credit scores. What does this imply about lenders' willingness to take...
-
The latest methodology adopted in auditing is the risk-based audit methodologyIn the light of the above statement, answer the following: (a) What is risk-based audit methodology? (b) Why audit risk...
-
Develop a checklist of items to consider in development of the site plan for either the bridge project or the building project.
-
A trader is worried that in spite of substantial increase in sales compared to the earlier year, there is considerable fall in gross profit. After satisfying himself that sales and expenses are...
-
What are some of the reasons schedules need to be shortened?
-
What is the importance of the schedule of values in project financial management?
-
Presented below is an aging schedule for Sycamore AG. At December 31, 2017, the unadjusted balance in Allowance for Doubtful Accounts is a credit of ¬9,200. Instructions (a) Journalize and post...
-
How much more interest will be earned if $5000 is invested for 6 years at 7% compounded continuously, instead of at 7% compounded quarterly?
-
What is the composition of the vapor phase in equilibrium with a liquid phase chloroform(1)/tetrahydrofuran(2) mixture of the following compositions at P = 1 bar? (a) x 1 = 0.1 (b) x 1 = 0.2 (c) x 1...
-
What is the composition of the liquid phase in equilibrium with a vapor phase ethanol(1)/ethyl acetate(2) mixture of the following compositions at P = 1 bar? (a) y 1 = 0.1 (b) y 1 = 0.2 (c) y 1 = 0.3...
-
What is the composition of the vapor phase in equilibrium with a liquid phase ethanol(1)/ethyl acetate(2) mixture of the following compositions at P = 1 bar? (a) x 1 = 0.1 (b) x 1 = 0.2 (c) x 1 = 0.3...
-
Dogs R Us reported a profit margin of 10.40 percent, total asset turnover of 0.74 times, debt-to-equity of 0.70 times, net income of $490,000, and dividends paid to common stockholders of $294,000....
-
5 Choose the correct word or phrase to complete the conversation. de moda recomiendas buena idea apretada opinion creo queda bien me parece que Alma: ;Es recomiendas ir de compras hoy? Migdalia: ; de...
-
The common stock of Devina Company is currently selling for $ 4 0 per share. It is expected that Devina will pay a dividend equal to $ 3 . 7 5 per share this year, D 1 . In addition, analyses have...

Study smarter with the SolutionInn App


