For an electron in the hydrogen state find (r) after first expressing it in terms of a
Question:
For an electron in the hydrogen state
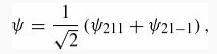
find (r) after first expressing it in terms of a single reduced matrix element.
Transcribed Image Text:
4 = 1 금 (V211 + 21-1),
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The expectation value of r is and applying Eq...View the full answer

Answered By

Amar Kumar Behera
I am an expert in science and technology. I provide dedicated guidance and help in understanding key concepts in various fields such as mechanical engineering, industrial engineering, electronics, computer science, physics and maths. I will help you clarify your doubts and explain ideas and concepts that are otherwise difficult to follow. I also provide proof reading services. I hold a number of degrees in engineering from top 10 universities of the US and Europe.
My experience spans 20 years in academia and industry. I have worked for top blue chip companies.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introduction To Quantum Mechanics
ISBN: 9781107189638
3rd Edition
Authors: David J. Griffiths, Darrell F. Schroeter
Question Posted:
Students also viewed these Engineering questions
-
A hydrogen atom has an electron in the n = 5 level. (a) If the electron returns to the ground state by emitting radiation, what is the minimum number of photons that can be emitted? (b) What is the...
-
An excited hydrogen atom with an electron in the n = 5 state emits light having a frequency of 6.90 1014 s-1. Determine the principal quantum level for the final state in this electronic transition.
-
For an electron in the ground state of a hydrogen atom, calculate its (a) Potential energy, (b) Kinetic energy, and (c) Total energy.
-
Find reactions of support 1,2 and 3 F=10KN, a=2m, ?a=60 (45%) F 2a
-
A survey is taken to estimate the mean annual family income for families living in public housing in Chicago. For a random sample of 29 families, the annual incomes (in hundreds of dollars) are as...
-
A ball of mass m and another ball of mass 3m are placed inside a smooth metal tube with a massless spring compressed between them. When the spring is released, the heavier ball flies out of one end...
-
Find the percentage in the following: 1. Total is 1,000 , percentage of the total is 70 . 2. Total is 500 , percentage of the total is 425 .
-
Physical Phitness, Inc. operates three divisions, Weak, Average, and Strong. As it turns out, the Weak division has the lowest operating income, and the president wants to close it. "Survival of the...
-
Paul Kwan owns a rental property that he acquired some years ago for $780,000. Of this total, $280,000 was allocated to the land and $500,000 was allocated to the building. An appraiser has estimated...
-
Aman and Juanita have $150,000 in their business bank account. You have calculated their warehouse stock and equipment to be valued at a total of $480,000. They want to take out a policy for...
-
Work out pH (t) for the system in Example 6.7 and comment on the correspondence with the classical equation of motion. Example 6.7 A particle of mass m moves in one dimension in a harmonic-oscillator...
-
Show that the commutator [L,f] = 0 leads to the same rule, Equation 6.46, as does the commutator [L,f] = 0. (n'lm| lm) = (n'l (m + 1) | (m +1)). (6.46)
-
In problem use appropriate algebra and Theorem 7.2.1 to find the given inverse Laplace transform. Theorem 7.2.1 s + 1 L- |s + 2 -1,
-
Sanfillipo, Inc., had 800 units of inventory on hand at March 1, 2006, costing $20 each. Purchases and sales of inventory during the month of March were as follows: Date Purchases March 8 Sales 600...
-
Which educational program would be most effective in reducing the body weight of overweight or obese children? Educating parents on the risks of childhood obesity Demonstration on cooking and...
-
Kirpalani Limited wants to expand its furniture line. The board of directors have agreed that the fastest way to raise these funds is to issue bonds. The company proceeds to issue $ 6 0 0 , 0 0 0 , 1...
-
introduction Overall, Steve Edwards, vice president of Marketing at Ditten-hoefers Fine China, is very pleased with the success of his new line of Gem-Surface china plates. Gem-Surface plates are...
-
Write a Press Release Imagine that you are about to open a new gym for teenagers. To generate publicity, you want to send the media a press release. Use a search engine to find out the standard...
-
Prove directly that the stiffness matrices in Examples 6.1 and 6.2 are positive definite.
-
Can partitioned join be used for r r.A s? Explain your answer
-
A foundation measuring 1.2 m 2.4 m in plan is constructed in a saturated clay. Given: depth of embedment of the foundation = 2 m, unit weight of soil = 18 kN/m 3 , and undrained cohesion of clay =...
-
Four point loads with the same magnitude of P are applied as shown in the plan view in Figure P8.1 and are separated by distance b. a. Find the vertical stress increase at a depth of 0.5b below the...
-
A point load of 500 kN is applied at the ground level. Plot the lateral variation of the vertical stress increase at depths of 2 m, 3 m, and 4 m below the ground level.
-
m(x) = csc(3x) - 1 is increasing on (, ) ...()u(0)(.) U... ...U (3,0) U(0, 1) U ( 3 O...U()()()u... U ... () (0) (,)..... U 3 none
-
The general environment analysis considers broad societal dimensions influencing the industry of firms within it. The general environment areas include? A. Political, Economic, Social, Technical,...
-
Dolan Company's accounting records reflect the following inventories: Dec. 31, 2022 Dec. 31, 2021 Raw materials $310,000 $260,000 inventory Work in process 300,000 160.000 inventory Finished goods...

Study smarter with the SolutionInn App


