At a given temperature, pressure, and initial composition, the equilibrium mole fraction of species C for the
Question:
At a given temperature, pressure, and initial composition, the equilibrium mole fraction of species C for the following reaction:![]()
is found to be 0.75. What is the value of the mole fraction if the reaction is written as follows?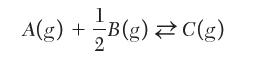
Transcribed Image Text:
2A(g) + B(g) 20(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
At low to moderate pressures the equilibrium state of the watergas shift reaction CO + H 2 O = CO 2 + H 2 is approximately described by the relation where T is the reactor temperature. K e is the...
-
A binary system of species l and 2 consists of vapor and liquid phase* in equilibrium at temperature T. The overall mole fraction of species I in the sy stem is d = 0.65. At temperature T. In 1 =...
-
The reaction N2 + O2 2NO occurs in internal combustion engines. Determine the equilibrium mole fraction of NO when the pressure is 101 kPa and the temperature is 1800 K.
-
There are 38 numbers in the game of roulette. They are 00, 0, 1, 2, . . ., 36. Each number has an equal chance of being selected. In the game, the winning number is found by a spin of the wheel. Say...
-
No major observatory has a Newtonian reflector as its primary instrument, whereas Newtonian reflectors are extremely popular among amateur astronomers. Explain why this is so.
-
Find the acute angle for each of the given trigonometric functions. Round off results according to Table 4.1. csc = 2.574 Data from Table 4.1 Angles and Accuracy of Trigonometric Functions...
-
Your local newsagent makes sales of 52,000 and purchases of 31,000 over the last accounting year. The owner took goods costing 455 without paying for them. Closing stock was valued at its cost of...
-
Primrose Beauty Supplies Ltd. was incorporated in January 2016. Primrose had the following transactions in its first month: Jan. 1 Received $150,000 in exchange for issuing 15,000 common shares. 1...
-
QUESTION 30 A project has estimated annual net cash flows of $46,000. It is estimated to cost $385,000. Determine the cash payback.. Round to 1 decimal point. 8.8 years 4.2 years 8.4 years 6.9 years
-
At 300 K and 1 bar, the equilibrium constant for the following reaction: is reported to be 10. What is the equilibrium constant at 300 K and 10 bar? You may assume ideal gas behavior. A(g) + B(g) C(g)
-
Consider the following reaction: The Gibbs energy of reaction at 298 K is determined to be and at a given temperature, the equilibrium constant is reported to be KT = 16. Now consider the reaction is...
-
Verify the indicated definition in Problems 1518 from Table 3.9 using a truth table. p unless q Table 3.9 Either p or q pq (pvq)^~(p^q) TT F TF FT FF T T F Neither p nor q p unless q ~(p vq) ~q p F T...
-
You Doll & Doll has two facilities that source, procure raw material, manufacture, QAQC and initial Pack for delivery. The company distributes the toys from these two plants through two distribution...
-
What is a 529 plan? What is a Coverdell Education Savings Account? What is a mutual fund expense ratio? What is a load fee? What is a no-load fund? What is a 12b-1 fee?
-
identify one redundant data in the table and explain why it is redundant (D) identify one insertion anomaly and explain it(E) identify 3 full functional dependencies and state your assumptions Name:...
-
2. A sample of 2 tsp of sugar, C12H22O11, weighs 9.00 grams. a. How many moles of sugar are in this sample: Show work here: Molar mass of sugar: (12 x 12) + (1 x 22) + (16x11)=(144 +22+176) g/mol...
-
Putting aside the question of tax credits, why might a government (whether federal or state) want to have regulated exchanges where individuals can purchase health insurance?
-
The following December 31, 2011, fiscal year-end account balance information is available for the Stonebridge Corporation: The only asset not listed is short-term investments. The only liabilities...
-
The Zwatch Company manufactures trendy, high-quality moderately priced watches. As Zwatch's senior financial analyst, you are asked to recommend a method of inventory costing. The CFO will use your...
-
One of these isomeric alcohols has mp = 26C and bp = 82C; the other has mp = 90C and hp = 117C. Explain which isomer has the higher melting point and which has the higher boiling point.
-
Explain the differences in the boiling points between the members of each of these pairs of compounds: (a) CH 3 (CH 2 )6 CH3 bp: 126C CH3 (CH2)8 CH3 bp: 174C (b) CH3CH2CH2OH bp: 97C CH3CH2OCH3 bp:...
-
Explain the difference in the melting points of these isomers: mp=-140C 0 mp = 7C
-
_______________: The seller agrees to accept a percentage of the sales price and stays on to manage the business for a few more years under the new owner; the remaining portion of the price is...
-
Private, not-for-profit colleges and universities must have a Statement of Financial Position, Statement of Activities, Statement of Cash Flows, and Notes to the Financial Statements included in...
-
You have just been hired into a management position which requires the application of your budgeting skills. You find out that budgeting has not been a priority of the company. You have contacted...

Study smarter with the SolutionInn App


