The molar enthalpy of a ternary mixture of species a, b, and c can be described by
Question:
The molar enthalpy of a ternary mixture of species a, b, and c can be described by the following expression: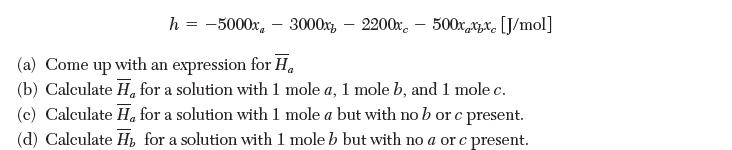
Transcribed Image Text:
h = -5000x - 3000x2200x500xxx [J/mol] (a) Come up with an expression for H (b) Calculate H for a solution with 1 mole a, 1 mole b, and 1 mole c. (c) Calculate H, for a solution with 1 mole a but with no b or c present. (d) Calculate H for a solution with 1 mole b but with no a orc present.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
Lets tackle each part of the question step by step a To come up with an expression for Ha the partia...View the full answer

Answered By

Pranav Makode
I am a bachelor students studying at professor ram meghe institute of technology and research. I have a great experience of being an expert. I have worked as an expert at helloexperts and solvelancer as a part time job. I have also worked as a doubt solver at ICAD SCHOOL OF LEARNING, which is in Amravati city. I have also worked as an Freelancer.
I have great experience of helping students, as described above. I can help any students in a most simple and understandable way. I will not give you have any chance for complaint. You will be greatfull to accept me as an expert.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Use the following information to calculate the net cash provided or used by financing activities. (a) Paid $28,200 cash to settle long-term notes payable at its $28,200 maturity value. (b) Acquired...
-
1. The molar enthalpy of a ternary mixture of species a, b, and c can be described by the following expression: a. hm = -5000xa - 3000xb - 2200xc-500xaxbxc [J/mol] b. Come up with an expression for...
-
The molar enthalpy of a binary liquid mixture of species 1 and 2 is given by: where T is the temperature in [K]. This expression is valid in the temperature range from 280 K to 360 K. Please answer...
-
Examine the difficulty of adding a proposed lwi.drd, rsl, rs2 (Load With Increment) instruction to MIPS. Interpretation: Reg[rd] = Mem[Reg[rs1] + Reg[rs2]] 1. Which new functional blocks (if any) do...
-
Write a paper that responds to the following: Compare and contrast the legal concepts of "reasonable suspicion" and "probable cause." How does each of these concepts apply to law enforcement...
-
Determine each of the following as being either true or false. If it is false, explain why. The circumference of a circle of diameter d is d.
-
Your employer, a large MNC, has asked you to assess its transaction exposure. Its projected cash flows are as follows for the next year. Danish krone inflows equal DK50,000,000 while outflows equal...
-
Haddas Ltd. makes Healthy Life, a nutritional aid. For a 50-pound batch, standard material and labor costs are as follows: During June, the following materials and labor were used in producing 600...
-
Evaluate the indefinite integral: (82-3 (8z +5z + 4z) dz=+C. +421)
-
Plot the partial molar volumes of CO2 and C3H8 in a binary mixture at 100C and 20 bar as a function of mole fraction CO2 using the van der Waals equation of state.
-
Consider a mixture of species 1, 2, and 3. The following equation of state is available for the vapor phase: where, and y1, y2, and y3 are the mole fractions of species 1, 2 and 3, respectively....
-
Solve the following equation. 3x = 5x + 1 - 1
-
i) What are the possible genotypes of the offspring of a cross between an Aa male parent x Aa female parent? Fill in Punnett Square below with the possible genotypes of the gametes and offspring....
-
Bonus money totalling $920,000 is to be shared by Dave, Claude, Marcel, and Larry by the ratio 15 : 11 : 8 : 6. How much will Dave receive?
-
Ellen purchases a 20% partnership interest but does not participate in the partnerships business. She owns no other passive investments. Her Section 704(d) basis in the partnership = $80,000. Of this...
-
If someone was a witness to a murder and they gave a police statement but before trial the defendant killed the witness what rule would be more applicable in regards to hearsay CEC rule 1241 or rule...
-
Consider the following Go program allowing the parallel processing of two slices of an array. Here, synchronization is achieved with a WaitGroup. Rewrite the program is performing, this time, a...
-
On the last day of its fiscal year ending December 31, 2011, the Sedgwick & Reams (S&R) Glass Company completed two financing arrangements. The funds provided by these initiatives will allow the...
-
The Home Depot is the leading retailer in the home improvement industry and one of the 10largest retailers in the United States. The company included the following on its January 29, 2012, balance...
-
Describe the mechanism of fluorescence. To what extent is a fluorescence spectrum not the exact mirror image of the corresponding absorption spectrum
-
Describe the principles of laser action, with actual examples.
-
The molar absorption coefficient of a substance dissolved in hexane is known to be 327 dm3 mol-1 cm-1 at 300 nm calculate the percentage reduction in intensity when light of that wavelength passes...
-
Required information Research Case 10-2 (Static) FASB codification; locate and extract relevant information and cite authoritative support for a financial reporting issue; restoration costs; asset...
-
Assets Cash Bowers Corporation Comparative Balance Sheets December 31, 20x2 and 20x1 ($ in thousands) Accounts receivable Short-term investment Inventory Land Buildings and equipment Less:...
-
Dwight Donovan, the president of Franklin Enterprises, is considering two investment opportunities. Because of limited resources, he will be able to invest in only one of them. Project A is to...

Study smarter with the SolutionInn App


