A student draws the diagram below and includes some reactions to represent the diagram. NaNO3 J +
Question:
A student draws the diagram below and includes some reactions to represent the diagram.
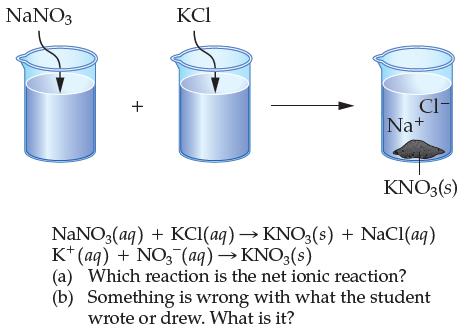
Transcribed Image Text:
NaNO3 J + KCI 00 CI- Na+ KNO3(s) NaNO3 (aq) + KCl(aq) → KNO3(s) + NaCl(aq) K+ (aq) + NO3 (aq) →KNO3(s) (a) Which reaction is the net ionic reaction? (b) Something is wrong with what the student wrote or drew. What is it?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
NaNOKC a b KNO4 NaCl NaNOKCKNONaC There are ...View the full answer

Answered By

Ashish Bhalla
I have 12 years work experience as Professor for Accounting, Finance and Business related subjects also working as Online Tutor from last 8 years with highly decentralized organizations. I had obtained a B.Com, M.Com, MBA (Finance & Marketing). My research interest areas are Banking Problem & Investment Management. I am highly articulate and effective communicator with excellent team-building and interpersonal skills; work well with individuals at all levels.
4.80+
17+ Reviews
46+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
A student is asked to sketch a pV diagram for a gas that goes through a cycle consisting of (a) An isobaric expansion, (b) A constant-volume reduction in temperature, (c) An isothermal process that...
-
A student is asked to sketch a pV diagram for a gas that goes through a cycle consisting of (a) an isobaric expansion, (b) a constant-volume reduction in temperature, and (c) an isothermal process...
-
Given the diagram below and knowing that point E lies on side AC, and point D lies on BE. If BC = n and DB = n 1 for some positive integer n and ABC = ACB = CED = CDE. Find the number of positive...
-
Find the area between the parabolas y = 2x 2 + 1 and y = x 2 + 5.
-
The Vest School of Vocational Technology has organized the school training programs into three departments. Each department provides training in a different area as follows: nursing assistant, dental...
-
The estimator of A in Exercise 17. If you think about the definition of the expectation, you might be able to demonstrate that this estimator is unbiased. Write down the equations that would express...
-
Which measure is useful in evaluating the efficiency in managing inventories? (a) Inventory turnover ratio. (b) Days in inventory. (c) Both (a) and (b). (d) None of the above.
-
Assume that your organization is planning to have a server room that functions without human beingsin other words, the functions are automated (such a room is often called a lights-out server room)....
-
Question 3 of 8 View Policies < > Show Attempt History Current Attempt in Progress Blossom Company issued $510,000, 10-year, 7% bonds at 102. (a) 0.88/2 Your answer is partially correct. Prepare a...
-
Using molecular formulas, write a balanced equation for the following gas-phase reaction. Then translate the balanced reaction into an English sentence. H" H H + Cl-Cl H H H Cl +H-Cl Cl
-
Write the complete ionic equation and net ionic equation for the neutralization reaction between aqueous hydrobromic acid and barium hydroxide.
-
Solve each equation. -x - 3x - 8 = 0
-
? Read this article to see what WSJ is saying about the Dove commercial: Byron, Ellen (Feb. 13, 2013). Marketing Decoder |Dove men + Care. The Wall Street Journal . Retrieved July 22, 2014 from:http...
-
What was your thoughts after watching the Khan video about deficits and debt? Did that help you understand the difference between the national debt and the deficit spending we engage in year after...
-
Considering the five P's- physicians, patients, payers, the public and politics. How have each generally changed? How do these changes impact healthcare marketing endeavors?
-
What does leadership do well in criminal justice and public safety?
-
Select an item that you purchased last month and that you had planned to purchase again this month. Now suppose its price increases by over 50%. Explain whether or not you would still purchase the...
-
A professor finds that she awards a final grade of A to 20% of her students. Of those who obtain a final grade of A, 70% obtained an A on the midterm examination. Also, 10% of the students who failed...
-
Borrowing costs should be recognised as an expense and charged to the profit and loss account of the period in which they are incurred : A. If the borrowing costs relate to qualifying asset B. If the...
-
The velocity of flow in a ventilation duct is 1140 ft/min. Compute the velocity in m/s.
-
A duct in a heating system carries 8320 cfm. Compute the flow rate in m 3 /s.
-
A pipe in a compressed air system carries 2650 cfm. Compute the flow rate in m 3 /s.
-
A single-step income statement shows only one subtotal for expenses.True false question.TrueFalse
-
Agito and its subsidiaries (Agito Group) are principally engaged in the production and distribution of high-density polyethylene (HDPE) in Hong Kong. On 1 April 2022, Agito signed an agreement with...
-
Problem 10-14 (Algo) Measures of Internal Business Process Performance [LO10-3] DataSpan, Incorporated, automated its plant at the start of the current year and installed a flexible manufacturing...

Study smarter with the SolutionInn App


