Assign an oxidation state to each atom in the amino acid glycine: - H-N-C-C T
Question:
Assign an oxidation state to each atom in the amino acid glycine:
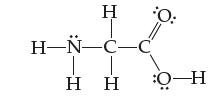
Transcribed Image Text:
Η Η- H-N-C-C T Η Η O-H 0:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
The following table shows the oxidation states of each atom in the amino acid glycine Atom Oxidation state H 1 C carbonyl carbon 1 C alpha carbon 1 N ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
In carbon dioxide, CO 2 , (a) Assign an oxidation state to each atom in the molecule. (b) How many electrons does the C atom own by oxidation-state electron bookkeeping? (c) How many more or fewer...
-
In chloroform, CHCl 3 , (a) Assign an oxidation state to each atom in the molecule. (b) How many electrons does the C atom own by oxidation-state electron bookkeeping? (c) How many more or fewer...
-
In methane, CH 4 , (a) Assign an oxidation state to each atom in the molecule. (b) How many electrons does the C atom own by oxidation-state electron bookkeeping? (c) How many more or fewer valence...
-
Problem 5-47 Amortizing Loans and Inflation (LO3) Suppose you take out a $108,000, 20-year mortgage loan to buy a condo. The interest rate on the loan is 5%. To keep things simple, we will assume you...
-
The comparative balance sheet of Putnam Cycle Co. at December 31, 2010 and 2009, is as follows: The noncurrent asset, noncurrent liability, and stockholders equity accounts for 2010 are as follows:...
-
What shapes would you expect for the following demand curves? a. A medicine that means life or death for a patient b. French fries in a food court with kiosks offering many types of food
-
What is the amount of gross profit for the month as shown on the income statement?
-
For Eckstein Company, the predetermined overhead rate is 130% of direct labor cost. During the month, Eckstein incurred $100,000 of factory labor costs, of which $85,000 is direct labor and $15,000...
-
y-values 9 8 g(x) 7 9 5 4 3 2 1 1 2 g(x) If f(x)= then h(x)' f'(3) 3 5 x-values S 4 h(x) 2 y-values 3 1 3 4 5 2 x-values
-
Identify the oxidizing agent and reducing agent in the reaction IO + 71 + 8 H412 + 4 HO
-
When you turn on an electrical appliance, are you consuming electrons?
-
A soft drink bottling company just ran a long line of 12-ounce soft drink cans filled with cola. A sample of 32 cans is selected by inspectors looking for nonconforming items. Among the things the...
-
Logistics Solutions provides order fulfillment services for dot.com merchants. The company maintains warehouses that stock items carried by its dot.com clients. When a client receives an order from a...
-
Name 3 characteristics, actions, or behaviors of a "good" employee; describe or give an example of the behavior. Be specific. Next, name 3 characteristics, actions or behaviors of the "bad" employee;...
-
1 . Falcon Corporation operates a widget manufacturing firm in a small town in Ohio. Falcon s sells its products primarily in the states of Ohio, Indiana, Michigan and Pennsylvania, making up 8 0 %...
-
Describe one thing you learned about globalization (positive analysis, "what is"). How do you feel about globalization given what you read for today (normative analysis, "what should be")? How would...
-
Suppose list is an array of 6 components of the type int. What is stored in the array list after the following Java code executes? list[0] = 5; for (int i=1; 6; i++) { listi +5; if (i > 2) list[i] =...
-
From the following accounts of Kurlz Salon, Inc., prepare the businesss statement of retained earnings for the year ended December 31, 2013: Retained Earnings Dividends Clo 95,000 Jan1 188,000 Mar 31...
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
A current source in a linear circuit has i s = 15 cos (25 t + 25) A (a) What is the amplitude of the current? (b) What is the angular frequency? (c) Find the frequency of the current. (d) Calculate...
-
Given v 1 = 45 sin(t + 30) V and v 2 = 50 cos(t 30) V, determine the phase angle between the two sinusoids and which one lags the other.
-
Transform the following sinusoids to phasors: (a) 20 cos(4t + 135) (b) 8 sin(20t + 30) (c) 20 cos (2t) + 15 sin (2t)
-
The records of Hoffman Company reflected the following balances in the stockholders equity accounts at December 31, 2018: Common stock, par $12 per share, 40,000 shares outstanding. Preferred stock,...
-
Problem 9-12 Direct Materials and Direct Labor Budgets [LO4, LO5] The production department of Prison Company has submitted the following forecast of units to be produced by quarter for the upcoming...
-
Exercise 10-6 (Algo) Contrasting Return on Investment (ROI) and Residual Income [LO10-1, LO10-2] Meiji Isetan Corporation of Japan has two regional divisions with headquarters in Osaka and Yokohama....

Study smarter with the SolutionInn App


