Convert this excited boron, B, atom to (a) An excited state of lower energy (b) The
Question:
Convert this excited boron, B, atom to
(a) An excited state of lower energy
(b) The ground state:
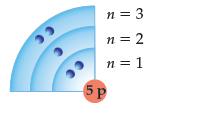
Transcribed Image Text:
33 СС i сл р n = 3 n=2 n = 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 28% (7 reviews)
a An excited state of lower energy To convert the excited boron atom in the image to an excited state of lower energy we need to move one of the elect...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
An atom in an excited state will on the average undergo a transition to a state of lower energy in about 10 -8 seconds. If the electron in a doubly ionized lithium atom (Li +2 , which is...
-
A transition of particular importance in 02 gives rise to the 'Schumann-Rung c band' in the ultraviolet region. The wave numbers (in cm-1) of transitions from the ground state to the vibrational...
-
An electron in a hydrogen atom is excited from the ground state to the n = 4 state. Comment on the correctness of the following statements (true or false). (a) n = 4 is the first excited state. (b)...
-
What does it mean that flame is non-luminous it is yellow it is invisible It has a very bright blue inner cone it is dark
-
What is a standard? Give some examples such as those appearing in the chapter introduction.
-
Explain the difference between zero, incomplete, and complete crowding out. If crowding out is complete, does it call into question the effectiveness of a rise in government purchases in order to...
-
What is a concentration account?
-
Can a creditor of both the husband and wife under the same obligation obtain an execution against a Winnebago mobile home owned by the husband and wife in tenancy by entirety?
-
Solve the equation on the interval [0, 2T). COS 2 x + 2 cos x + 1 = 0 3 O 1,37 22 OTT 2TT
-
According to Bohrs model, is energy absorbed or released when an electron moves to a shell of lower n?
-
Would moving an electron farther from an atoms nucleus give off light energy or require the absorption of light energy? Explain your answer.
-
Complete the following MRP Record for item A: ITEM: A LT: 2 Lot Size: 250 PERIOD 6 Gross requirements Scheduled receipts Projected on hand 120 Net requirements Planned order releases 100 150500...
-
The Fall-Out Shelter Problem: You are a member of a department in Washington D.C., which oversees experimental stations in the outposts of civilization. Suddenly the Third World War breaks out and...
-
According to anthropologist Edward T . Hall, which social interaction zone describes communication that usually occurs at approximately 1 . 5 to 4 feet? Explain
-
Summers corporation recently used $75,000 of direct materials and $9,000 of indirect materials in production activities. What would be the journal entries reflecting these transactions?
-
It is January 1 of Year 2 . Company sales for January, February, and March are forecasted as follows: January: $ 2 0 , 0 0 0 February: $ 4 0 , 0 0 0 March: $ 5 0 , 0 0 0 Seventy percent of sales are...
-
Eight years ago, Raj Hapi launched Hapi's Petting Zoo & Caf (Hapi's). Hapi's is a not-for-profit zoo located in the Annapolis Valley in Nova Scotia. It is a place where people can interact with...
-
Construct a 95% confidence interval to estimate the population mean using the following data: x = 38 = 8.5 n = 25 What assumptions need to be made about this population?
-
Refer to the situation described inBE 18-13, but assume a 2-for-1 stock split instead of the 5% stock dividend. Prepare the journal entry to record the stock split if it is to be effected in the form...
-
There are more electrons in the n = 4 shell than for the n = 3 shell in krypton. However, the peak in the radial distribution in Figure 21.6 is smaller for the n = 4 shell than for the n = 3 shell....
-
How is the effective nuclear charge related to the size of the basis set in a Hartree Fock calculation?
-
Using the result of Problem P20.12, calculate the probability of finding the electron in the 1s state outside a sphere of radius 0.75a 0 , 2.5a 0 , and 4.5a 0 .
-
ABC Costing Method what is the the "Using Gross Profit %"for the 3 empty grey spaces ... Note the existing profit is not 52.17% for the second empty box Question 1 The sustainability manager is...
-
A health savings plan Question 14 options: a) may be an involuntary payroll deduction for an employee. b) allows users to save money that is deducted after taxes to pay for future medical expenses....
-
Madison Manufacturing wants to import a machine from Germany. However, the company's head of manufacturing is reluctant to spend the time required to obtain a letter of credit that the German...

Study smarter with the SolutionInn App


