Indicate with an arrow the direction of the equilibrium shift and predict what will happen to the
Question:
Indicate with an arrow the direction of the equilibrium shift and predict what will happen to the amount of Fe2O3 (increases, decreases, unchanged, need more information) when the following stresses are applied to the following exothermic reaction:
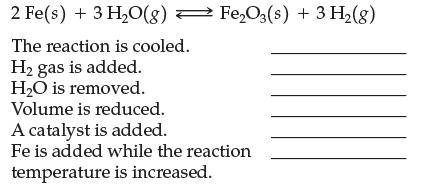
Transcribed Image Text:
2 Fe(s) + 3 H₂O(g)Fe₂O3(s) + 3 H₂(g) The reaction is cooled. H₂ gas is added. H₂O is removed. Volume is reduced. A catalyst is added. Fe is added while the reaction temperature is increased.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Direction of equilibrium shift Cooling The reaction is exothermic so cooling the system will shift t...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Predict what will happen to the equilibrium price and quantity of oranges if the following events take place. (L03) a. A study finds that a daily glass of orange juice reduces the risk of heart...
-
Predict what will happen to aggregate output and the price level if the Federal Reserve increases the money supply at the same time that Congress implements and income tax cut.
-
Predict what will happen to interest rates if prices in the bond market become more volatile.
-
Consider the nutrition problem in Example 1 of Section 3.3. Solve the problem by the simplex method, and then determine the optimal quantities of soybeans and rice in the diet, and the new cost, if...
-
Determining Financial Statement Effects of Sales and Cost of Goods Sold and Issuance of Par Value Stock Using the following categories, indicate the effects of the following transactions. Use + for...
-
Most university athletic programs now have a Web site with information on individual sports and a Prospective Student Athlete Form that allows high school athletes to submit their academic and sports...
-
describe how currency futures contracts are used to speculate or hedge based on anticipated exchange rate movements, and
-
The equity categories for Swire Pacific Limited are illustrated in Exhibit 13-9, on page 801. Using the information in the exhibit, answer the following questions: 1. Recall that the primary purpose...
-
Op plify each of the following and w (5+ 14)(1 + 12i
-
How does decreasing the temperature affect the value of K eq for an exothermic reaction? (a) Increases K eq (b) Decreases K eq (c) Does not change K eq
-
Shown below is a concentration versus time plot for the reaction A B. Using the data on the graph, determine the equilibrium constant for the reaction. Concentration (M)- 0.100 0.080 0.060 0.040...
-
What are three examples of intentional threats?
-
Although many people do not immediately recognize it, many people enter into contracts each and every day. The healthcare profession in particular is loaded with contractual relationships, and...
-
Captain Shea Brennan discussed selling his home and 20 acres to Thomas for $225,000. Thomas wanted a couple of days to think about buying the land and home. James Dutton had learned that Captain Shea...
-
Clara was hired on a four-month fixed-term contract to perform crisis response work, including responding to calls from area hospitals to assess and provide support for persons who presented as being...
-
Price = $25 Variable Cost= $15 Fixed Cost= $130 000 Tax Rate= 30% Compute cm Compute cm ratio Compute be in units assume that the company was able to reduce the VC per unit by $3. What selling price...
-
In contrast, futures contracts, with their liquidity and transparent pricing, provide an efficient means for traders and investors to hedge against market fluctuations. The requirement for margin...
-
Franco, Corp. completed the following transactions in 2014: Jan 1 Purchased a building costing $140,000 and signed a 10-year, 11 % mortgage note payable for the same amount. Jun 30 Made the first...
-
In 1995 Miguel purchased a home for $130,000. In 2000 he sold it for $170,000 and immediately purchased another one for $180,000, which he sold in 2007 for $235,000. How much taxable capital gain, if...
-
A bicycle is moving initially with a constant velocity along a level road. The bicyclist then decides to slow down, so she applies her brakes over a period of several seconds. Thereafter, she again...
-
Make a qualitative sketch of the position y as a function of time for the center of a yo-yo (the point at the middle of the axle). Also make sketches of the velocity and acceleration as functions of...
-
In SI units, velocity is measured in units of meters per second (m/s). Which of the following combinations of units can also be used to measure velocity? (a) Cm/s (b) Cm/s 2 (c) m 3 /(mm 2 s 2 ) (d)...
-
Why are revenue and expense accounts called temporary? Are there other temporary accounts
-
1.Kathy Morris owns a small decorating business called Premium Decorating. A partiallycompleted worksheet for Premium Decorating and additional information follow directly.Additional Information1....
-
If compliance with the continuing education requirement is by self-attestation, proof of participation in required continuing education is the responsibility of the licensee. To ensure that proof of...

Study smarter with the SolutionInn App


