The acetate ion has the formula C 2 H 3 O 2 . Draw a dot
Question:
The acetate ion has the formula C2H3O2–. Draw a dot diagram for it given the following atomic connections.
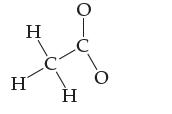
Transcribed Image Text:
H Η 0-6 Ο C Η
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
H C HCC 0 ...View the full answer

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Acetaldehyde has the formula C 2 H 4 O. Draw a dot diagram for it given the following atomic connections. H C 0- C
-
Acetic acid, a major component of vinegar, has the formula C 2 H 4 O 2 . Draw a dot diagram for it given the following atomic connections. H C C 0-H
-
Ethanol, the alcohol found in alcoholic beverages, has the same formula as the ether of Problem 5.78. However, it has a different structure, in which only one carbon atom is bound to the oxygen atom....
-
PROJECT SCENARIO You are recently appointed as the IT Consultant and Software Architect in BMS a Software Architect firm. As your first project, you are assigned by the Project Manager to propose and...
-
Materials often represent a substantial portion of a companys assets; therefore, they should be controlled from the time orders are placed to the time finished goods are shipped to the customer. What...
-
Give two examples of political or legal forces and explain how they might interact with economic forces
-
Why can convertible bonds be placed so quickly?
-
The Foreman Companys earnings and common stock dividends have been growing at an annual rate of 6 percent over the past 10 years and are expected to continue growing at this rate for the foreseeable...
-
(a) There are 13 European cities that Joe would eventually like to visit. On his next vacation, though, he only has time to visit 3 of the cities: one on Monday, one on Tuesday, and one on Wednesday....
-
Draw a dot diagram for perchloric acid, HClO 4 . The chlorine is the central atom to which all the oxygens are attached, and the hydrogen is attached to one of the oxygens.
-
Draw a dot diagram for the hypothetical O 2 2+ ion.
-
The Excel worksheet form that appears below is to be used to recreate an example earlier in the chapter. Download the workbook containing this form from Connect. On the website you will also receive...
-
Based on a predicted level of production and sales of 37,000 units, a company anticipates total contribution margin of $151,700, fixed costs of $74,000, and income of $77,700. Based on this...
-
A total of 35 hp is required for a particular pumping application. If the pump efficiency is 85%, what is the brake horsepower required
-
Have the worldwide (globalization) demand for quality driven the concept of innovation and how has innovation change life. Explain your answer in at least 6 statements.
-
BERRAPCALCBR7 1.4.065. 12. [0/1 Points] DETAILS For the function, find and simplify f(x) = 6x - 8x +1 PREVIOUS ANSWERS f(x+h) f(x) (Assume h + 0.) h +4xh+2h - 8x 8h+1 f(x + h) - f(x) = h 6x2 Need...
-
plz answer b and c Activity 5.1 The following cost and activity data were recorded for the Long Mountain Dispensary for the last two months ended 30 June 2004. In Patients April June 1400 1700...
-
The following data show the number of pounds of bananas sold per week at the Avalon Grocery store when the banana display was positioned in the produce, milk, and cereal sections of the store. These...
-
Research corporate acquisitions using Web resources and then answer the following questions: Why do firms purchase other corporations? Do firms pay too much for the acquired corporation? Why do so...
-
Answer the following questions using the PT phase diagram for carbon sketched below. a. Which substance is denser, graphite or diamond? Explain your answer. b. Which phase is more dense, graphite or...
-
You have a compound dissolved in chloroform and need to remove the solvent by distillation. Because the compound is heat sensitive, you hesitate to raise the temperature above 5.00C and decide on...
-
Why is it reasonable to show the μ versus T segments for the three phases as straight lines as is done in Figure 8.1? More realistic curves would have some curvature. Is the curvature...
-
All costs in a homogeneous cost pool have Question 60 options: A) only one cost driver that will accurately represent the relationship between the costs and the activity. B) to be adjusted at year...
-
Which of the following is true for B-corps? Question 3 options: a) It is a for-profit corporation. b) It is a nonprofit corporation. c) They are taxed differently than c-corporations. d) They are...
-
Because interest earned on U.S. bonds and notes is not taxed by Oregon, special treatment is required if expenses related to this income is deducted as an itemized expense on your federal return....

Study smarter with the SolutionInn App


